OR

Anidulafungin
V-Echinocandin
| CAS Number | 166663-25-8 |
|---|---|
N-[(3S,6S,9S,11R,15S,18S,20R,21R,24S,25S,26S)-6-[(1S,2R)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl]-11,20,21,25-tetrahydroxy-3,15-bis[(1R)-1-hydroxyethyl]-26-methyl-2,5,8,14,17,23-hexaoxo-1,4,7,13,16,22-hexaazatricyclo[22.3.0.09,13]heptacosan-18-yl]- 4-{4-[4-(pentyloxy)phenyl]phenyl}benzamide
- LY-307853
- LY-329960
- LY-333006
- LY303366
- VEC
- VER-002
1H NMR (700 MHz, d6-DMSO) δ 0.91 (t, 3H), 1.12 (d, 3H), 1.36 (m, 2H), 1.41 (m, 2H), 1.74 (p, 2H), 1.88 and 1.97 (overlapped, 2H), 3.85 (overlapped, 1H), 4.01 (t, 2H), 4.35 (overlapped, 1H), 4.44 (m, 1H), 4.76 (m, 1H), 4.80 (m, 1H), 5.02 (m, 1H), 5.07 (d, 1H), 5.52 (d, 1H), 7.04 (d, 1H), 7.66 (d, 1H), 7.74 (d, 1H), 7.80 (d, 1H), 7.82 (d, 1H), 7.97 (d, 1H), 8.01 (d, 1H), 8.14 (broad s, 1H), 8.60 (d, 1H). IR (cm−1)
KBr νmax; 3450 (O−H), 2932 (C−H), 2871 (C−H), 1632 (C═O), 1517 (Ar), 1488 (Ar), 1248 (C−O), 821 (C−H out-of-plane bending Ar 2 adj H’s).
Anidulafungin (brand names: Eraxis (in U.S. and Russia), Ecalta (in Europe)) is a semisynthetic echinocandin used as anantifungal drug. Anidulafungin was originally manufactured and submitted for FDA approval by Vicuron Pharmaceuticals.[1] Pfizeracquired the drug upon its acquisition of Vicuron in the fall of 2005.[2] Pfizer gained approval by the Food and Drug Administration(FDA) on February 21, 2006;[3] it was previously known as LY303366. Preliminary evidence indicates it has a similar safety profile tocaspofungin. Anidulafungin has proven efficacy against esophageal candidiasis, but its main use will probably be in invasive Candidainfection;[4][5][6] it may also have application in treating invasive Aspergillus infection. It is a member of the class of antifungal drugs known as the echinocandins; its mechanism of action is by inhibition of (1→3)-β-D-glucan synthase, an enzyme important to the synthesis of the fungal cell wall.
Pharmacodynamics and pharmacokinetics
Anidulafungin significantly differs from other antifungals in that it undergoes chemical degradation to inactive forms at body pH and temperature. Because it does not rely on enzymatic degradation or hepatic or renal excretion, the drug is safe to use in patients with any degree of hepatic or renal impairment.[7]
Distribution: 30–50 L. Protein binding: 84%.
Anidulafungin is not evidently metabolized by the liver. This specific drug undergoes slow chemical hydrolysis to an open-ring peptide which lacks antifungal activity. The half-life of the drug is 27 hours. Thirty percent is excreted in the feces (10% as unchanged drug). Less than 1% is excreted in the urine.[8][9][10]
Mechanism of action
Anidulafungin inhibits glucan synthase, an enzyme important in the formation of (1→3)-β-D-glucan, a major fungal cell wall component. Glucan synthase is not present in mammalian cells, so it is an attractive target for antifungal activity.[11]
Semisynthesis
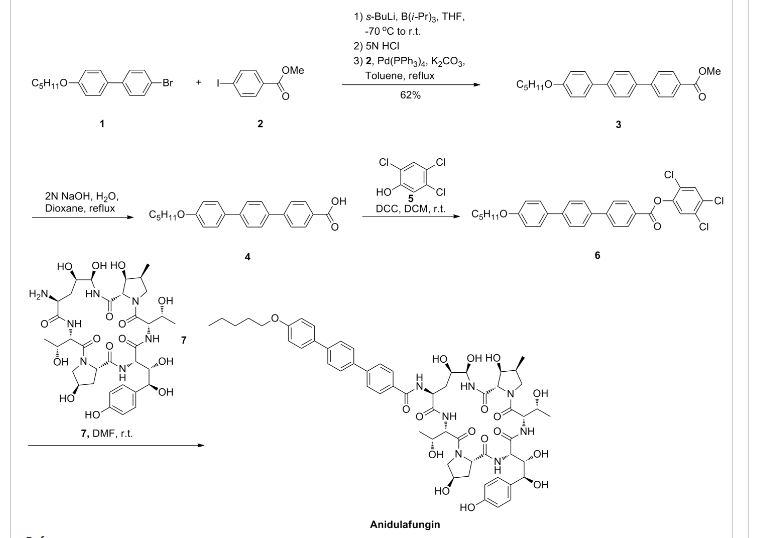
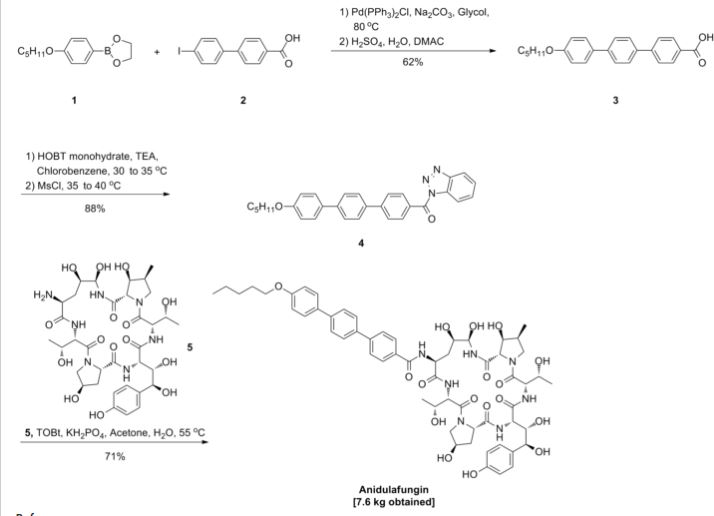
Anidulafungin is manufactured via semisynthesis. The starting material is echinocandin B (a lipopeptide fermentation product ofAspergillus nidulans or the closely related species, A. rugulosus), which undergoes deacylation (cleavage of the linoleoyl side chain) by the action of a deacylase enzyme from the bacterium Actinoplanes utahensis;[12] in three subsequent synthetic steps, including a chemical reacylation, the antifungal drug anidulafungin[11][13] is synthesized.
Aspergillus nidulans. Anidulafungin is an echinocandin, a class of antifungal drugs that inhibits the synthesis of 1,3-β-D-glucan, an essential component of fungal cell walls.
ERAXIS (anidulafungin) is 1-[(4R,5R)-4,5-dihydroxy-N -[[4“-(pentyloxy)[1,1':4',1”-terphenyl]-4-yl]carbonyl]-L-ornithine]echinocandin B. Anidulafungin is a white to off-white powder that is practically insoluble in water and slightly soluble in ethanol. In addition to the active ingredient, anidulafungin, ERAXIS for Injection contains the following inactive ingredients:
50 mg/vial - fructose (50 mg), mannitol (250 mg), polysorbate 80 (125 mg), tartaric acid (5.6 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
100 mg/vial - fructose (100 mg), mannitol (500 mg), polysorbate 80 (250 mg), tartaric acid (11.2 mg), and sodium hydroxide and/or hydrochloric acid for pH adjustment.
The empirical formula of anidulafungin is C58H73N7O17 and the formula weight is 1140.3. The structural formula is

Prior to administration, ERAXIS for Injection requires reconstitution with sterile Water for Injection and subsequent dilution with either 5% DextroseInjection, USP or 0.9% Sodium Chloride Injection, USP (normal saline).
SYNTHESIS
J MED CHEM 1995, 38 3271-3281
Semisynthetic Chemical Modification of the Antifungal Lipopeptide ...
pubs.acs.org/doi/abs/10.1021/jm00017a012
by M Debono - 1995 - Cited by 113 - Related articles
Aug 1, 1995 - J. Med. Chem. , 1995, 38 (17), pp 3271–3281. DOI: 10.1021/jm00017a012 ... Journal ofMedicinal Chemistry 2001 44 (16), 2671-2674
Echinocandin B (ECB) is a lipopeptide composed of a complex cyclic peptide acylated at the N-terminus by linoleic acid. Enzymatic deacylation of ECB provided the peptide "nucleus" as a biologically inactive substrate from which novel ECB analogs were generated by chemical reacylation at the N-terminus. Varying the acyl group revealed that the structure and physical properties of the side chain, particularly its geometry and lipophilicity, played a pivotal role in determining the antifungal potency properties of the analog. Using CLOGP values to describe and compare the lipophilicities of the side chain fragments, it was shown that values of > 3.5 were required for expression of antifungal activity. Secondly, a linearly rigid geometry of the side chain was the most effective shape in enhancing the antifungal potency. Using these parameters as a guide, a variety of novel ECB analogs were synthesized which included arylacyl groups that incorporated biphenyl, terphenyl, tetraphenyl, and arylethynyl groups. Generally the glucan synthase inhibition by these analogs correlated well with in vitro and in vivo activities and was likewise influenced by the structure of the side chain. These structural variations resulted in enhancement of antifungal activity in both in vitro and in vivo assays. Some of these analogs, including LY303366 (14a), were effective by the oral route of administration.
PATENT
US 5965525
PATENT
US 4293482
Paper
Commercialization and Late-Stage Development of a Semisynthetic Antifungal API: Anidulafungin/d-Fructose (Eraxis)
Chemical Research and Development, Pfizer Inc. Global Research and Development Laboratories, Eastern Point Road, Groton, Connecticut 06340, U.S.A.
Org. Process Res. Dev., 2008, 12 (3), pp 447–455
DOI: 10.1021/op800055h
* Corresponding author. E-mail: timothy.norris@pfizer.com. Telephone: +860 441 4406 . Fax: +860 686 5340.

Many years ago anidulafungin 1 was identified as a potentially useful medicine for the treatment of fungal infections. Its chemical and physical properties as a relatively high molecular weight semisynthetic derived from echinocandin B proved to be a significant hurdle to its final presentation as a useful medicine. It has recently been approved as an intravenous treatment for invasive candidaisis, an increasingly common health hazard that is potentially life-threatening. The development and commercialization of this API, which is presented as a molecular mixture of anidulafungin and d-fructose is described. This includes, single crystal X-ray structures of the starting materials, the echinocandin B cyclic-peptide nucleus (ECBN·HCl) and the active ester 1-({[4′′-(pentyloxy)-1,1′:4′,1′′-terphenyl-4-yl]carbonyl}oxy)-1H-1,2,3-benzotriazole (TOBt). Details of the structure and properties of starting materials, scale-up chemistry and unusual crystallization phenomena associated with the API formation are discussed.
References
- PRNewswire. Vicuron Pharmaceuticals Files New Drug Application (NDA) for Anidulafungin for Treatment of Invasive Candidiasis/Candidemia 08-18-2005.
- Jump up^ PRNewswire. Vicuron Pharmaceuticals Stockholders Approve Merger With Pfizer 08-15-2005
- "FDA Approves New Treatment for Fungal Infections". FDA News Release. Food and Drug Administration. 2006-02-21. Archived from the original on 10 July 2009. Retrieved 2009-08-01.
- Krause DS, Reinhardt J, Vazquez JA, Reboli A, Goldstein BP, Wible M, Henkel T (2004). "Phase 2, randomized, dose-ranging study evaluating the safety and efficacy of anidulafungin in invasive candidiasis and candidemia". Antimicrob Agents Chemother 48 (6): 2021–4.doi:10.1128/AAC.48.6.2021-2024.2004. PMC 415613. PMID 15155194.
- Jump up^ Pfaller MA, Boyken L, Hollis RJ, Messer SA, Tendolkar S, Diekema DJ (2005). "In Vitro Activities of Anidulafungin against More than 2,500 Clinical Isolates of Candida spp., Including 315 Isolates Resistant to Fluconazole". J Clin Microbiol 43 (11): 5425–7.doi:10.1128/JCM.43.11.5425-5427.2005. PMC 1287823. PMID 16272464.
- J Pfaller MA, Diekema DJ, Boyken L, Messer SA, Tendolkar S, Hollis RJ, Goldstein BP (2005). "Effectiveness of anidulafungin in eradicating Candida species in invasive candidiasis". Antimicrob Agents Chemother 49 (11): 4795–7. doi:10.1128/AAC.49.11.4795-4797.2005.PMC 1280139. PMID 16251335.
- Jump up^ "Eraxis at RxList". 2009-06-24. Retrieved 2009-08-01.
- Trissel LA and Ogundele AB, "Compatibility of Anidulafungin With Other Drugs During Simulated Y-Site Administration,"Am J Health-Sys Pharm, 2005, 62:834-7.
- Vazquez JA, "Anidulafungin: A New Echinocandin With a Novel Profile," Clin Ther, 2005, 27(6):657-73.
- Jump up^ Walsh TJ, Anaissie EJ, Denning DW, et al., "Treatment of Aspergillosis: Clinical Practice Guidelines of the Infectious Diseases Society of America," Clin Infect Dis, 2008, 46(3):327-60
- Denning DW (1997). "Echinocandins and pneumocandins - a new antifungal class with a novel mode of action". J Antimicrob Chemother 40 (5): 611–614. doi:10.1093/jac/dkf045.PMID 9421307.
- Lei Shao; Jian Li; Aijuan Liu; Qing Chang; Huimin Lin; Daijie Chen (2013). "Efficient Bioconversion of Echinocandin B to Its Nucleus by Overexpression of Deacylase Genes in Different Host Strains". Applied and Environmental Microbiology 79 (4): 1126–1133. doi:10.1128/AEM.02792-12. PMC 3568618. PMID 23220968.
- "Anidulafungin EMA Europa" (PDF).
 | |
| Systematic (IUPAC) name | |
|---|---|
| N-[(3S,6S,9S,11R,15S,18S,20R,21R,24S,25S,26S)-6-[(1S,2R)-1,2-dihydroxy-2-(4-hydroxyphenyl)ethyl]-11,20,21,25-tetrahydroxy-3,15-bis[(1R)-1-hydroxyethyl]-26-methyl-2,5,8,14,17,23-hexaoxo-1,4,7,13,16,22-hexaazatricyclo[22.3.0.09,13]heptacosan-18-yl]- 4-{4-[4-(pentyloxy)phenyl]phenyl}benzamide | |
| Clinical data | |
| Trade names | Eraxis |
| AHFS/Drugs.com | Monograph |
| Pharmacokinetic data | |
| Protein binding | 84 % |
| Biological half-life | 40–50 hours |
| Identifiers | |
| CAS Number | 166663-25-8 |
| ATC code | J02AX06 (WHO) |
| PubChem | CID 166548 |
| DrugBank | DB00362 |
| ChemSpider | 21106258 |
| UNII | 9HLM53094I |
| KEGG | D03211 |
| ChEBI | CHEBI:55346 |
| ChEMBL | CHEMBL1630215 |
| Chemical data | |
| Formula | C58H73N7O17 |
| Molar mass | 1140.24 g/mol |
//////////FUNGIN, ANIDULAFUNGIN, Eraxis , Ecalta, semisynthetic echinocandin, anantifungal drug, FDA 2006, PFIZER, LY-307853, LY-329960, LY-333006, LY303366, VEC, VER-002, 166663-25-8, Eli Lilly and Company Inc.
CCCCCOc1ccc(cc1)c2ccc(cc2)c3ccc(cc3)C(=O)N[C@H]6C[C@@H](O)[C@@H](O)NC(=O)C4[C@@H](O)[C@@H](C)CN4C(=O)C(NC(=O)C(NC(=O)C5C[C@@H](O)CN5C(=O)C(NC6=O)[C@@H](C)O)[C@@H](O)[C@H](O)c7ccc(O)cc7)[C@@H](C)O