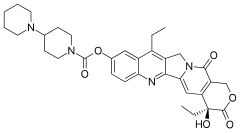
squalamine
July 10, 2013 /
Ohr Pharmaceutical, Inc. , a pharmaceutical company focused on the development of novel therapeutics for large unmet medical needs, today announced that it has enrolled the first 60 patients in the Company's ongoing OHR-002 Phase II clinical trial evaluating Squalamine Eye Drops for the treatment of the wet form of age-related macular degeneration ("wet-AMD").
The study is a randomized, double blind, placebo controlled study enrolling patients at more than twenty clinical sites in the U.S.
"We are excited to have reached the halfway point in the enrollment of our Squalamine Eye Drop study," said Dr. Irach B. Taraporewala, CEO of Ohr.
"This milestone sets the stage for the planned interim analysis once the 60 patients complete the nine month treatment protocol, and we expect the data to be available in the second quarter of 2014.
"
"I am pleased with the continued progress in the trial to evaluate Squalamine Eye Drops for exudative AMD," commented Dr. Jason Slakter, retinal disease specialist at Vitreous-Retina-Macula Consultants of NY, and member of Ohr's scientific advisory board. "The Company's eye drop for treating wet-AMD would potentially offer patients a convenient, self-administered, treatment alternative or adjunct to currently used intravitreal injections directly into the eye. This could be a significant advancement in the future treatment of wet-AMD."
Study OHR-002 is a randomized, double blind, placebo controlled Phase II study to evaluate the efficacy and safety of Squalamine Eye Drops for the treatment of wet-AMD.
The study will enroll 120 treatment naive wet-AMD patients at more than twenty clinical sites in the U.S., who will be treated with Squalamine Eye Drops or placebo eye drops twice daily for a nine month period.
The primary and secondary endpoints include visual acuity parameters, need for rescue intravitreal injections, and safety. The protocol includes an interim analysis upon the completion of the treatment period in 50% of the patients (60). More information on the clinical trial can be found at clinicaltrials.gov.
About Ohr Pharmaceutical, Inc.
Ohr Pharmaceutical, Inc. (NasdaqCM:OHRP) is a pharmaceutical company dedicated to the clinical development of new drugs for underserved therapeutic needs in large and growing markets. The Company is focused on advancing its pipeline products currently in phase II clinical development: Squalamine Eye Drops for the treatment of the wet form of age-related macular degeneration, and OHR/AVR118 for the treatment of cancer cachexia. Additional information on the Company can be found at www.ohrpharmaceutical.com.