Printing with Collagen
Addition of collagen to hydrogels in 3D printing improves stem cell differentiation in osteogenesis
- Bioprinting Organotypic Hydrogels with Improved Mesenchymal Stem Cell Remodeling and Mineralization Properties for Bone Tissue Engineering,
Daniela Filipa Duarte Campos, Andreas Blaeser, Kate Buellesbach, Kshama Shree Sen, Weiwei Xun, Walter Tillmann, Horst Fischer,
Adv. Healthcare Mater. 2016.
DOI: 10.1002/adhm.201501033
MORE ON
Collagen

Tropocollagen molecule: three left-handed procollagens (red, green, blue) join to form a right handed triple helical tropocollagen.

Collagen is the most common protein found in mammals.

Collagen /ˈkɒlədʒᵻn/ is the main structural protein in the extracellular space in the various connective tissues in animal bodies. As the main component of connective tissue, it is the most abundant protein in mammals,[1] making up from 25% to 35% of the whole-body protein content. Depending upon the degree of mineralization, collagen tissues may be rigid (bone), compliant (tendon), or have a gradient from rigid to compliant (cartilage).[2] Collagen, in the form of elongated fibrils, is mostly found in fibrous tissues such as tendons, ligaments and skin. It is also abundant incorneas, cartilage, bones, blood vessels, the gut, intervertebral discs and the dentin in teeth.[3] In muscle tissue, it serves as a major component of the endomysium. Collagen constitutes one to two percent of muscle tissue, and accounts for 6% of the weight of strong, tendinous muscles.[4] Thefibroblast is the most common cell that creates collagen.
Gelatin, which is used in food and industry, is collagen that has been irreversibly hydrolyzed.[5] Collagen also has many medical uses in treating complications of the bones and skin.
The name collagen comes from the Greek κόλλα (kólla), meaning "glue", and suffix -γέν, -gen, denoting "producing".[6][7] This refers to the compound's early use in the process of boiling the skin and sinews of horses and other animals to obtain glue.

Collagen injections can be used in cosmetic procedures to improve the contours of aging skin.
Types of collagen
Collagen occurs in many places throughout the body. Over 90% of the collagen in the human body, however, is type I.[8]
So far, 28 types of collagen have been identified and described. They can be divided into several groups according to the structure they form:[2]
- Fibrillar (Type I, II, III, V, XI)
- Non-fibrillar
- FACIT (Fibril Associated Collagens with Interrupted Triple Helices) (Type IX, XII, XIV, XVI, XIX)
- Short chain (Type VIII, X)
- Basement membrane (Type IV)
- Multiplexin (Multiple Triple Helix domains with Interruptions) (Type XV, XVIII)
- MACIT (Membrane Associated Collagens with Interrupted Triple Helices) (Type XIII, XVII)
- Other (Type VI, VII)
The five most common types are:
- Type I: skin, tendon, vascular ligature, organs, bone (main component of the organic part of bone)
- Type II: cartilage (main collagenous component of cartilage)
- Type III: reticulate (main component of reticular fibers), commonly found alongside type I.
- Type IV: forms basal lamina, the epithelium-secreted layer of the basement membrane.
- Type V: cell surfaces, hair and placenta

Tobacco contains chemicals that damage collagen
Medical uses
Cardiac applications
The collagenous cardiac skeleton which includes the four heart valve rings, is histologically and uniquely bound to cardiac muscle. The cardiac skeleton also includes the separating septa of the heart chambers – the interventricular septum and the atrioventricular septum. Collagen contribution to the measure of cardiac performance summarily represents a continuous torsional force opposed to the fluid mechanics of blood pressure emitted from the heart. The collagenous structure that divides the upper chambers of the heart from the lower chambers is an impermeable membrane that excludes both blood and electrical impulses through typical physiological means. With support from collagen, atrial fibrillation should never deteriorate to ventricular fibrillation. Collagen is layered in variable densities with cardiac muscle mass. The mass, distribution, age and density of collagen all contribute to the compliance required to move blood back and forth. Individual cardiac valvular leaflets are folded into shape by specialized collagen under variable pressure. Gradual calcium deposition within collagen occurs as a natural function of aging. Calcified points within collagen matrices show contrast in a moving display of blood and muscle, enabling methods of cardiac imaging technology to arrive at ratios essentially stating blood in (cardiac input) and blood out (cardiac output). Pathology of the collagen underpinning of the heart is understood within the category of connective tissue disease.
Hydrolyzed type II collagen and osteoarthritis
A published study[9] reports that ingestion of a novel low molecular weight hydrolyzed chicken sternal cartilage extract, containing a matrix of hydrolyzed type II collagen,chondroitin sulfate, and hyaluronic acid, relieves joint discomfort associated with osteoarthritis. A randomized controlled trial (RCT) enrolling 80 subjects demonstrated that it was well tolerated with no serious adverse event and led to a significant improvement in joint mobility compared to the placebo group on days 35 (p = 0.007) and 70 (p < 0.001).
Fast facts on collagen
Here are some key points about collagen. More detail and supporting information is in the main article.25-27
- Protein makes up around 20% of the body's mass, and collagen makes up around 30% of the protein in the human body.
- There are at least 16 types of collagen, but 80-90% of the collagen in the body consists of types I, II, and III.
- Type I collagen fibrils are stronger than steel (gram for gram).
- Collagen is most commonly found within the body in the skin, bones and connective tissues.
- The word "collagen" is derived from the Greek "kolla," meaning glue.
- Collagen gives the skin its strength and structure, and also plays a role in the replacement of dead skin cells.
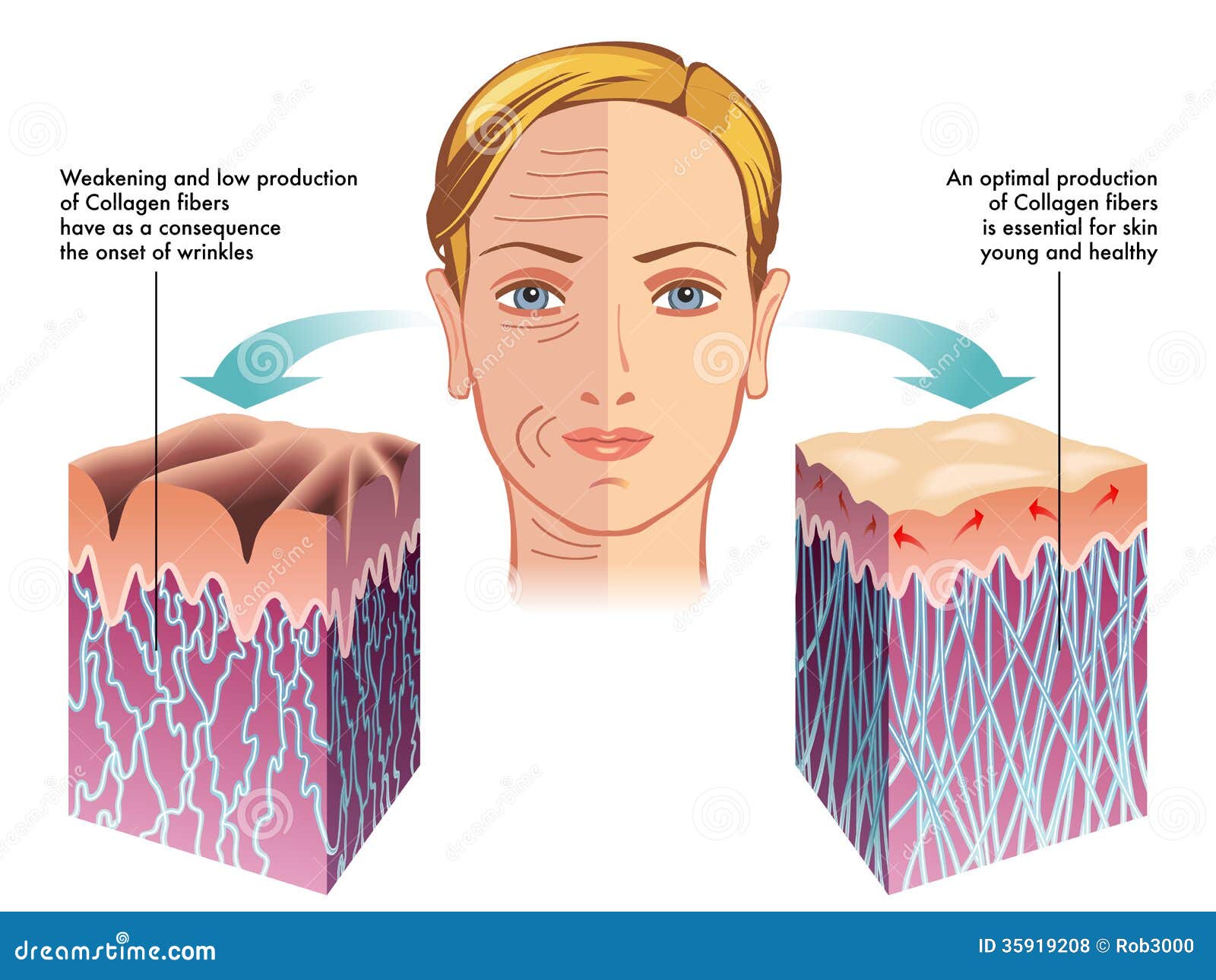
- Collagen production declines with age (as part of intrinsic aging), and is reduced by exposure to ultraviolet light and other environmental factors (extrinsic aging).
- Collagen in medical products can be derived from human, bovine, porcine and ovine sources.
- Collagen dressings attract new skin cells to wound sites.
- Cosmetic products such as revitalizing lotions that claim to increase collagen levels are unlikely to do so, as collagen molecules are too large to be absorbed through the skin.
- Collagen production can be stimulated through the use of laser therapy and the use of all-trans retinoic acid (a form ofvitamin A).
- Controllable factors that damage the production of collagen include sunlight, smoking and high sugar consumption.

Cosmetic surgery
Collagen has been widely used in cosmetic surgery, as a healing aid for burn patients for reconstruction of bone and a wide variety of dental, orthopedic, and surgical purposes. Both human and bovine collagen is widely used as dermal fillers for treatment of wrinkles and skin aging.[10] Some points of interest are:
- When used cosmetically, there is a chance of allergic reactions causing prolonged redness; however, this can be virtually eliminated by simple and inconspicuous patch testing prior to cosmetic use.
- Most medical collagen is derived from young beef cattle (bovine) from certified BSE-free animals. Most manufacturers use donor animals from either "closed herds", or from countries which have never had a reported case of BSE such as Australia, Brazil, and New Zealand.
Bone grafts
As the skeleton forms the structure of the body, it is vital that it maintains its strength, even after breaks and injuries. Collagen is used in bone grafting as it has a triple helical structure, making it a very strong molecule. It is ideal for use in bones, as it does not compromise the structural integrity of the skeleton. The triple helical structure of collagen prevents it from being broken down by enzymes, it enables adhesiveness of cells and it is important for the proper assembly of the extracellular matrix.[11]
Tissue regeneration
Collagen scaffolds are used in tissue regeneration, whether in sponges, thin sheets, or gels. Collagen has the correct properties for tissue regeneration such as pore structure, permeability, hydrophilicity and it is stable in vivo. Collagen scaffolds are also ideal for the deposition of cells, such as osteoblasts and fibroblasts and once inserted, growth is able to continue as normal in the tissue.[12]
Reconstructive surgical uses
Collagens are widely employed in the construction of the artificial skin substitutes used in the management of severe burns. These collagens may be derived from bovine, equine, porcine, or even human sources; and are sometimes used in combination with silicones, glycosaminoglycans, fibroblasts, growth factors and other substances.
Collagen is also sold commercially in pill form as a supplement to aid joint mobility. However, because proteins are broken down into amino acids before absorption, there is no reason for orally ingested collagen to affect connective tissue in the body, except through the effect of individual amino acid supplementation.
Collagen is also frequently used in scientific research applications for cell culture, studying cell behavior and cellular interactions with the extracellular environment.[13]
Wound care
Collagen is one of the body’s key natural resources and a component of skin tissue that can benefit all stages of the wound healing process.[14] When collagen is made available to the wound bed, closure can occur. Wound deterioration, followed sometimes by procedures such as amputation, can thus be avoided.
Collagen is a natural product, therefore it is used as a natural wound dressing and has properties that artificial wound dressings do not have. It is resistant against bacteria, which is of vital importance in a wound dressing. It helps to keep the wound sterile, because of its natural ability to fight infection. When collagen is used as a burn dressing, healthygranulation tissue is able to form very quickly over the burn, helping it to heal rapidly.[15]
Throughout the 4 phases of wound healing, collagen performs the following functions in wound healing:
- Guiding function: Collagen fibers serve to guide fibroblasts. Fibroblasts migrate along a connective tissue matrix.
- Chemotactic properties: The large surface area available on collagen fibers can attract fibrogenic cells which help in healing.
- Nucleation: Collagen, in the presence of certain neutral salt molecules can act as a nucleating agent causing formation of fibrillar structures. A collagen wound dressing might serve as a guide for orienting new collagen deposition and capillary growth.
- Hemostatic properties: Blood platelets interact with the collagen to make a hemostatic plug.
Chemistry
The collagen protein is composed of a triple helix, which generally consists of two identical chains (α1) and an additional chain that differs slightly in its chemical composition (α2).[16] The amino acid composition of collagen is atypical for proteins, particularly with respect to its high hydroxyproline content. The most common motifs in the amino acid sequence of collagen are glycine-proline-X and glycine-X-hydroxyproline, where X is any amino acid other than glycine, proline or hydroxyproline. The average amino acid composition for fish and mammal skin is given.[16]
| Amino acid | Abundance in mammal skin (residues/1000) | Abundance in fish skin (residues/1000) |
|---|---|---|
| Glycine | 329 | 339 |
| Proline | 126 | 108 |
| Alanine | 109 | 114 |
| Hydroxyproline | 95 | 67 |
| Glutamic acid | 74 | 76 |
| Arginine | 49 | 52 |
| Aspartic acid | 47 | 47 |
| Serine | 36 | 46 |
| Lysine | 29 | 26 |
| Leucine | 24 | 23 |
| Valine | 22 | 21 |
| Threonine | 19 | 26 |
| Phenylalanine | 13 | 14 |
| Isoleucine | 11 | 11 |
| Hydroxylysine | 6 | 8 |
| Methionine | 6 | 13 |
| Histidine | 5 | 7 |
| Tyrosine | 3 | 3 |
| Cysteine | 1 | 1 |
| Tryptophan | 0 | 0 |
Synthesis
First, a three-dimensional stranded structure is assembled, with the amino acids glycine and proline as its principal components. This is not yet collagen but its precursor, procollagen. Procollagen is then modified by the addition of hydroxyl groups to the amino acids proline and lysine. This step is important for later glycosylation and the formation of the triple helix structure of collagen. The hydroxylase enzymes that perform these reactions require Vitamin C as a cofactor, and a deficiency in this vitamin results in impaired collagen synthesis and the resulting disease scurvy[17] These hydroxylation reactions are catalyzed by two different enzymes: prolyl-4-hydroxylase[18] and lysyl-hydroxylase. Vitamin C also serves with them in inducing these reactions. In this service, one molecule of vitamin C is destroyed for each H replaced by OH. [19] The synthesis of collagen occurs inside and outside of the cell. The formation of collagen which results in fibrillary collagen (most common form) is discussed here. Meshwork collagen, which is often involved in the formation of filtration systems, is the other form of collagen. All types of collagens are triple helices, and the differences lie in the make-up of the alpha peptides created in step 2.
- Transcription of mRNA: About 34 genes are associated with collagen formation, each coding for a specific mRNA sequence, and typically have the "COL" prefix. The beginning of collagen synthesis begins with turning on genes which are associated with the formation of a particular alpha peptide (typically alpha 1, 2 or 3).
- Pre-pro-peptide formation: Once the final mRNA exits from the cell nucleus and enters into the cytoplasm, it links with the ribosomal subunits and the process of translation occurs. The early/first part of the new peptide is known as the signal sequence. The signal sequence on the N-terminal of the peptide is recognized by a signal recognition particle on the endoplasmic reticulum, which will be responsible for directing the pre-pro-peptide into the endoplasmic reticulum. Therefore, once the synthesis of new peptide is finished, it goes directly into the endoplasmic reticulum for post-translational processing. It is now known as pre-pro-collagen.
- Pre-pro-peptide to pro-collagen: Three modifications of the pre-pro-peptide occur leading to the formation of the alpha peptide:
- The signal peptide on the N-terminal is dissolved, and the molecule is now known as propeptide (not procollagen).
- Hydroxylation of lysines and prolines on propeptide by the enzymes 'prolyl hydroxylase' and 'lysyl hydroxylase' (to produce hydroxyproline and hydroxylysine) occurs to aid cross-linking of the alpha peptides. This enzymatic step requires vitamin C as a cofactor. In scurvy, the lack of hydroxylation of prolines and lysines causes a looser triple helix (which is formed by three alpha peptides).
- Glycosylation occurs by adding either glucose or galactose monomers onto the hydroxyl groups that were placed onto lysines, but not on prolines.
- Once these modifications have taken place, three of the hydroxylated and glycosylated propeptides twist into a triple helix forming procollagen. Procollagen still has unwound ends, which will be later trimmed. At this point, the procollagen is packaged into a transfer vesicle destined for the Golgi apparatus.
- Golgi apparatus modification: In the Golgi apparatus, the procollagen goes through one last post-translational modification before being secreted out of the cell. In this step, oligosaccharides (not monosaccharides as in step 3) are added, and then the procollagen is packaged into a secretory vesicle destined for the extracellular space.
- Formation of tropocollagen: Once outside the cell, membrane bound enzymes known as 'collagen peptidases', remove the "loose ends" of the procollagen molecule. What is left is known as tropocollagen. Defects in this step produce one of the many collagenopathies known as Ehlers-Danlos syndrome. This step is absent when synthesizing type III, a type of fibrilar collagen.
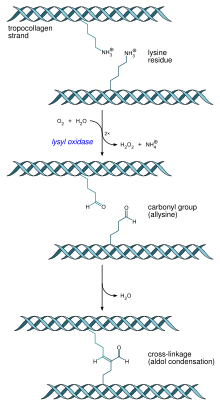
- Formation of the collagen fibril: 'Lysyl oxidase', an extracellular enzyme, produces the final step in the collagen synthesis pathway. This enzyme acts on lysines and hydroxylysines producing aldehyde groups, which will eventually undergo covalent bonding between tropocollagen molecules. This polymer of tropocollogen is known as a collagen fibril.
Amino acids
Collagen has an unusual amino acid composition and sequence:
- Glycine is found at almost every third residue.
- Proline makes up about 17% of collagen.
- Collagen contains two uncommon derivative amino acids not directly inserted during translation. These amino acids are found at specific locations relative to glycine and are modified post-translationally by different enzymes, both of which require vitamin C as acofactor.
- Hydroxyproline derived from proline
- Hydroxylysine derived from lysine - depending on the type of collagen, varying numbers of hydroxylysines are glycosylated (mostly having disaccharides attached).
Collagen I formation
Most collagen forms in a similar manner, but the following process is typical for type I:
- Inside the cell
- Two types of alpha chains are formed during translation on ribosomes along the rough endoplasmic reticulum (RER): alpha-1 and alpha-2 chains. These peptide chains (known as preprocollagen) have registration peptides on each end and a signal peptide.
- Polypeptide chains are released into the lumen of the RER.
- Signal peptides are cleaved inside the RER and the chains are now known as pro-alpha chains.
- Hydroxylation of lysine and proline amino acids occurs inside the lumen. This process is dependent on ascorbic acid (vitamin C) as a cofactor.
- Glycosylation of specific hydroxylysine residues occurs.
- Triple alpha helical structure is formed inside the endoplasmic reticulum from two alpha-1 chains and one alpha-2 chain.
- Procollagen is shipped to the Golgi apparatus, where it is packaged and secreted by exocytosis.
- Outside the cell
- Registration peptides are cleaved and tropocollagen is formed by procollagen peptidase.
- Multiple tropocollagen molecules form collagen fibrils, via covalent cross-linking (aldol reaction) by lysyl oxidase which links hydroxylysine and lysine residues. Multiple collagen fibrils form into collagen fibers.
- Collagen may be attached to cell membranes via several types of protein, including fibronectin and integrin.
Synthetic pathogenesis
Vitamin C deficiency causes scurvy, a serious and painful disease in which defective collagen prevents the formation of strong connective tissue. Gums deteriorate and bleed, with loss of teeth; skin discolors, and wounds do not heal. Prior to the 18th century, this condition was notorious among long-duration military, particularly naval, expeditions during which participants were deprived of foods containing vitamin C.
An autoimmune disease such as lupus erythematosus or rheumatoid arthritis[21] may attack healthy collagen fibers.
Many bacteria and viruses secrete virulence factors, such as the enzyme collagenase, which destroys collagen or interferes with its production.
Molecular structure
A single collagen molecule, tropocollagen, is used to make up larger collagen aggregates, such as fibrils. It is approximately 300 nm long and 1.5 nm in diameter, and it is made up of three polypeptide strands (called alpha peptides, see step 2), each of which has the conformation of a left-handed helix – this should not be confused with the right-handedalpha helix. These three left-handed helices are twisted together into a right-handed triple helix or "super helix", a cooperative quaternary structure stabilized by many hydrogen bonds. With type I collagen and possibly all fibrillar collagens, if not all collagens, each triple-helix associates into a right-handed super-super-coil referred to as the collagen microfibril. Each microfibril is interdigitated with its neighboring microfibrils to a degree that might suggest they are individually unstable, although within collagen fibrils, they are so well ordered as to be crystalline.
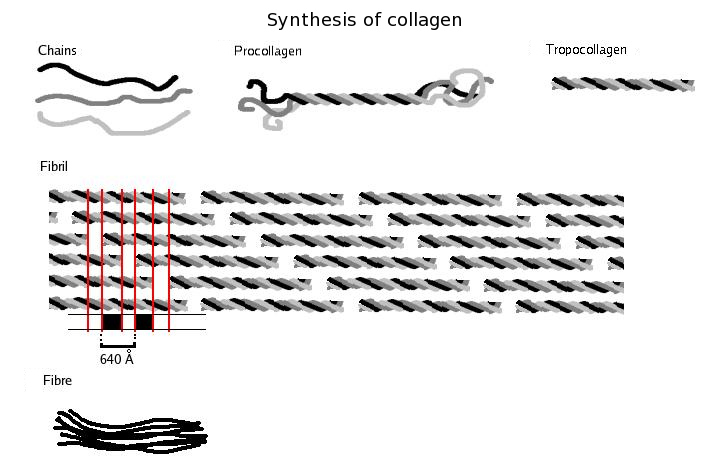
Three polypeptides coil to form tropocollagen. Many tropocollagens then bind together to form a fibril, and many of these then form a fibre.
A distinctive feature of collagen is the regular arrangement ofamino acids in each of the three chains of these collagen subunits. The sequence often follows the pattern Gly-Pro-X or Gly-X-Hyp, where X may be any of various other amino acid residues.[16] Proline or hydroxyproline constitute about 1/6 of the total sequence. With glycine accounting for the 1/3 of the sequence, this means approximately half of the collagen sequence is not glycine, proline or hydroxyproline, a fact often missed due to the distraction of the unusual GX1X2 character of collagen alpha-peptides. The high glycine content of collagen is important with respect to stabilization of the collagen helix as this allows the very close association of the collagen fibers within the molecule, facilitating hydrogen bonding and the formation of intermolecular cross-links.[16]This kind of regular repetition and high glycine content is found in only a few other fibrous proteins, such as silk fibroin.
Collagen is not only a structural protein. Due to its key role in the determination of cell phenotype, cell adhesion, tissue regulation and infrastructure, many sections of its non-proline-rich regions have cell or matrix association / regulation roles. The relatively high content of proline and hydroxyproline rings, with their geometrically constrained carboxyl and (secondary) amino groups, along with the rich abundance of glycine, accounts for the tendency of the individual polypeptide strands to form left-handed helices spontaneously, without any intrachain hydrogen bonding.
Because glycine is the smallest amino acid with no side chain, it plays a unique role in fibrous structural proteins. In collagen, Gly is required at every third position because the assembly of the triple helix puts this residue at the interior (axis) of the helix, where there is no space for a larger side group than glycine’s single hydrogen atom. For the same reason, the rings of the Pro and Hyp must point outward. These two amino acids help stabilize the triple helix—Hyp even more so than Pro; a lower concentration of them is required in animals such as fish, whose body temperatures are lower than most warm-blooded animals. Lower proline and hydroxyproline contents are characteristic of cold-water, but not warm-water fish; the latter tend to have similar proline and hydroxyproline contents to mammals.[16] The lower proline and hydroxproline contents of cold-water fish and other poikilotherm animals leads to their collagen having a lower thermal stability than mammalian collagen.[16] This lower thermal stability means that gelatin derived from fish collagen is not suitable for many food and industrial applications.
The tropocollagen subunits spontaneously self-assemble, with regularly staggered ends, into even larger arrays in the extracellular spaces of tissues.[22][23] Additional assembly of fibrils is guided by fibroblasts, which deposit fully formed fibrils from fibripositors.[2] In the fibrillar collagens, the molecules are staggered from each other by about 67 nm (a unit that is referred to as ‘D’ and changes depending upon the hydration state of the aggregate). Each D-period contains four plus a fraction collagen molecules, because 300 nm divided by 67 nm does not give an integer (the length of the collagen molecule divided by the stagger distance D). Therefore, in each D-period repeat of the microfibril, there is a part containing five molecules in cross-section, called the “overlap”, and a part containing only four molecules, called the "gap".[24] The triple-helices are also arranged in a hexagonal or quasihexagonal array in cross-section, in both the gap and overlap regions.[24][25]
There is some covalent crosslinking within the triple helices, and a variable amount of covalent crosslinking between tropocollagen helices forming well organized aggregates (such as fibrils).[26] Larger fibrillar bundles are formed with the aid of several different classes of proteins (including different collagen types), glycoproteins and proteoglycans to form the different types of mature tissues from alternate combinations of the same key players.[23] Collagen's insolubility was a barrier to the study of monomeric collagen until it was found that tropocollagen from young animals can be extracted because it is not yet fully crosslinked. However, advances in microscopy techniques (i.e. electron microscopy (EM) and atomic force microscopy (AFM)) and X-ray diffraction have enabled researchers to obtain increasingly detailed images of collagen structure in situ. These later advances are particularly important to better understanding the way in which collagen structure affects cell–cell and cell–matrix communication, and how tissues are constructed in growth and repair, and changed in development and disease.[27][28] For example, using AFM–based nanoindentation it has been shown that a single collagen fibril is a heterogeneous material along its axial direction with significantly different mechanical properties in its gap and overlap regions, correlating with its different molecular organizations in these two regions.[29]
Collagen fibrils/aggregates are arranged in different combinations and concentrations in various tissues to provide varying tissue properties. In bone, entire collagen triple helices lie in a parallel, staggered array. 40 nm gaps between the ends of the tropocollagen subunits (approximately equal to the gap region) probably serve as nucleation sites for the deposition of long, hard, fine crystals of the mineral component, which is (approximately) Ca10(OH)2(PO4)6.[30] Type I collagen gives bone its tensile strength.
Associated disorders
Collagen-related diseases most commonly arise from genetic defects or nutritional deficiencies that affect the biosynthesis, assembly, postranslational modification, secretion, or other processes involved in normal collagen production.
| Type | Notes | Gene(s) | Disorders |
| I | This is the most abundant collagen of the human body. It is present in scar tissue, the end product when tissue heals by repair. It is found in tendons, skin, artery walls, cornea, the endomysiumsurrounding muscle fibers, fibrocartilage, and the organic part of bones and teeth. | COL1A1, COL1A2 | Osteogenesis imperfecta, Ehlers–Danlos syndrome, Infantile cortical hyperostosis a.k.a. Caffey's disease |
| II | Hyaline cartilage, makes up 50% of all cartilage protein. Vitreous humour of the eye. | COL2A1 | Collagenopathy, types II and XI |
| III | This is the collagen of granulation tissue, and is produced quickly by young fibroblasts before the tougher type I collagen is synthesized. Reticular fiber. Also found in artery walls, skin, intestines and the uterus | COL3A1 | Ehlers–Danlos syndrome, Dupuytren's contracture |
| IV | Basal lamina; eye lens. Also serves as part of the filtration system in capillaries and the glomeruli ofnephron in the kidney. | COL4A1, COL4A2,COL4A3,COL4A4,COL4A5,COL4A6 | Alport syndrome, Goodpasture's syndrome |
| V | Most interstitial tissue, assoc. with type I, associated with placenta | COL5A1, COL5A2,COL5A3 | Ehlers–Danlos syndrome (Classical) |
| VI | Most interstitial tissue, assoc. with type I | COL6A1, COL6A2,COL6A3,COL6A5 | Ulrich myopathy, Bethlem myopathy,Atopic dermatitis[31] |
| VII | Forms anchoring fibrils in dermoepidermal junctions | COL7A1 | Epidermolysis bullosa dystrophica |
| VIII | Some endothelial cells | COL8A1, COL8A2 | Posterior polymorphous corneal dystrophy 2 |
| IX | FACIT collagen, cartilage, assoc. with type II and XI fibrils | COL9A1, COL9A2,COL9A3 | EDM2 and EDM3 |
| X | Hypertrophic and mineralizing cartilage | COL10A1 | Schmid metaphyseal dysplasia |
| XI | Cartilage | COL11A1, COL11A2 | Collagenopathy, types II and XI |
| XII | FACIT collagen, interacts with type I containing fibrils, decorin and glycosaminoglycans | COL12A1 | – |
| XIII | Transmembrane collagen, interacts with integrin a1b1, fibronectin and components of basement membranes like nidogen and perlecan. | COL13A1 | – |
| XIV | FACIT collagen, also known as undulin | COL14A1 | – |
| XV | – | COL15A1 | – |
| XVI | – | COL16A1 | – |
| XVII | Transmembrane collagen, also known as BP180, a 180 kDa protein | COL17A1 | Bullous pemphigoid and certain forms of junctional epidermolysis bullosa |
| XVIII | Source of endostatin | COL18A1 | – |
| XIX | FACIT collagen | COL19A1 | – |
| XX | – | COL20A1 | – |
| XXI | FACIT collagen | COL21A1 | – |
| XXII | – | COL22A1 | – |
| XXIII | MACIT collagen | COL23A1 | – |
| XXIV | – | COL24A1 | – |
| XXV | – | COL25A1 | – |
| XXVI | – | EMID2 | – |
| XXVII | – | COL27A1 | – |
| XXVIII | – | COL28A1 | – |
In addition to the above-mentioned disorders, excessive deposition of collagen occurs in scleroderma.
Diseases
One thousand mutations have been identified in twelve out of more than twenty types of collagen. These mutations can lead to various diseases at the tissue level.[32]
Osteogenesis imperfecta – Caused by a mutation in type 1 collagen, dominant autosomal disorder, results in weak bones and irregular connective tissue, some cases can be mild while others can be lethal, mild cases have lowered levels of collagen type 1 while severe cases have structural defects in collagen.[33]
Chondrodysplasias – Skeletal disorder believed to be caused by a mutation in type 2 collagen, further research is being conducted to confirm this.[34]
Ehlers-Danlos Syndrome – Six different types of this disorder, which lead to deformities in connective tissue. Some types can be lethal, leading to the rupture of arteries. Each syndrome is caused by a different mutation, for example type four of this disorder is caused by a mutation in collagen type 3.[35]
Alport syndrome – Can be passed on genetically, usually as X-linked dominant, but also as both an autosomal dominant and autosomal recessive disorder, sufferers have problems with their kidneys and eyes, loss of hearing can also develop in during the childhood or adolescent years.[36]
Osteoporosis – Not inherited genetically, brought on with age, associated with reduced levels of collagen in the skin and bones, growth hormone injections are being researched as a possible treatment to counteract any loss of collagen.[37]
Knobloch syndrome – Caused by a mutation in the COL18A1 gene that codes for the production of collagen XVIII. Patients present with protrusion of the brain tissue and degeneration of the retina, an individual who has family members suffering from the disorder are at an increased risk of developing it themselves as there is a hereditary link.[32]
Characteristics
Collagen is one of the long, fibrous structural proteins whose functions are quite different from those of globular proteins, such as enzymes. Tough bundles of collagen calledcollagen fibers are a major component of the extracellular matrix that supports most tissues and gives cells structure from the outside, but collagen is also found inside certain cells. Collagen has great tensile strength, and is the main component of fascia, cartilage, ligaments, tendons, bone and skin.[38][39] Along with elastin and soft keratin, it is responsible for skin strength and elasticity, and its degradation leads to wrinkles that accompany aging.[10] It strengthens blood vessels and plays a role in tissue development. It is present in the cornea and lens of the eye in crystalline form. It may be one of the most abundant proteins in the fossil record, given that it appears to fossilize frequently, even in bones from the Mesozoic and Paleozoic.[40]
Uses
Collagen has a wide variety of applications, from food to medical. For instance, it is used in cosmetic surgery and burn surgery. It is widely used in the form of collagen casings for sausages, which are also used in the manufacture of musical strings.
If collagen is subject to sufficient denaturation, e.g. by heating, the three tropocollagen strands separate partially or completely into globular domains, containing a different secondary structure to the normal collagen polyproline II (PPII), e.g. random coils. This process describes the formation of gelatin, which is used in many foods, including flavoredgelatin desserts. Besides food, gelatin has been used in pharmaceutical, cosmetic, and photography industries.[41] From a nutritional point of view, collagen and gelatin are a poor-quality sole source of protein since they do not contain all the essential amino acids in the proportions that the human body requires—they are not 'complete proteins' (as defined by food science, not that they are partially structured). Manufacturers of collagen-based dietary supplements usually containing hydrolyzed collagen claim that their products can improve skin and fingernail quality as well as joint health. However, mainstream scientific research has not shown strong evidence to support these claims.[42]Individuals with problems in these areas are more likely to be suffering from some other underlying condition (such as normal aging, dry skin, arthritis etc.) rather than just a protein deficiency.
From the Greek for glue, kolla, the word collagen means "glue producer" and refers to the early process of boiling the skin and sinews of horses and other animals to obtain glue. Collagen adhesive was used by Egyptians about 4,000 years ago, and Native Americans used it in bows about 1,500 years ago. The oldest glue in the world, carbon-dated as more than 8,000 years old, was found to be collagen—used as a protective lining on rope baskets and embroidered fabrics, and to hold utensils together; also in crisscross decorations on human skulls.[43] Collagen normally converts to gelatin, but survived due to dry conditions. Animal glues are thermoplastic, softening again upon reheating, and so they are still used in making musical instruments such as fine violins and guitars, which may have to be reopened for repairs—an application incompatible with tough, syntheticplastic adhesives, which are permanent. Animal sinews and skins, including leather, have been used to make useful articles for millennia.
Gelatin-resorcinol-formaldehyde glue (and with formaldehyde replaced by less-toxic pentanedial and ethanedial) has been used to repair experimental incisions in rabbit lungs.[44]
History
The molecular and packing structures of collagen have eluded scientists over decades of research. The first evidence that it possesses a regular structure at the molecular level was presented in the mid-1930s.[45][46] Since that time, many prominent scholars, including Nobel laureates Crick, Pauling, Rich and Yonath, and others, including Brodsky,Berman, and Ramachandran, concentrated on the conformation of the collagen monomer. Several competing models, although correctly dealing with the conformation of each individual peptide chain, gave way to the triple-helical "Madras" model of Ramachandran, which provided an essentially correct model of the molecule's quaternary structure[47][48][49] although this model still required some refinement.[50] [clarification needed] [51][52][53][54] The packing structure of collagen has not been defined to the same degree outside of the fibrillar collagen types, although it has been long known to be hexagonal or quasi-hexagonal.[25][55][56] As with its monomeric structure, several conflicting models alleged that either the packing arrangement of collagen molecules is 'sheet-like' or microfibrillar.[50][57][58] The microfibrillar structure of collagen fibrils in tendon, cornea and cartilage has been directly imaged by electron microscopy.[59][60][61] The microfibrillar structure of tail tendon, as described by Fraser, Miller, and Wess (amongst others), was modeled as being closest to the observed structure,[50] although it oversimplified the topological progression of neighboring collagen molecules, and hence did not predict the correct conformation of the discontinuous D-periodic pentameric arrangement termed simply: the microfibril.[24][62] Various cross linking agents like L-Dopaquinone, embeline, potassium embelate and 5-O-methyl embelin could be developed as potential cross-linking/stabilization agents of collagen preparation and its application as wound dressing sheet in clinical applications is enhanced.[63]
See also
- Hydrolyzed collagen, a common form in which collagen is sold as a supplement.
- Animal glue
- Gelatine
- Fibrous protein
- Osteoid, collagen containing component of bone
- Lysyl oxidase and LOXL1, LOXL2, LOXL3, LOXL4 in collagen formation
- Collagenase, the enzyme involved in collagen breakdown and remodelling. For more on other proteases that target collagen see The Proteolysis Map
- Ehlers-Danlos Syndrome
- Hypermobility Syndrome
- Sponge - Cell types
- Drug discovery and development of MMP inhibitors
References
- Jump up^ Di Lullo, Gloria A.; Sweeney, Shawn M.; Körkkö, Jarmo; Ala-Kokko, Leena & San Antonio, James D. (2002). "Mapping the Ligand-binding Sites and Disease-associated Mutations on the Most Abundant Protein in the Human, Type I Collagen". J. Biol. Chem.277 (6): 4223–4231. doi:10.1074/jbc.M110709200. PMID 11704682.
- ^ Jump up to:a b c Sherman, Vincent R. (2015). "The materials science of collagen". Journal of the Mechanical Behavior of Biomedical Materials 52: 22–50.doi:10.1016/j.jmbbm.2015.05.023.
- Jump up^ Britannica Concise Encyclopedia 2007
- Jump up^ Sikorski, Zdzisław E. (2001). Chemical and Functional Properties of Food Proteins. Boca Raton: CRC Press. p. 242. ISBN 1-56676-960-4.
- Jump up^ Bogue, Robert H. (1923). "Conditions Affecting the Hydrolysis of Collagen to Gelatin".Industrial and Engineering Chemistry 15 (11): 1154–1159. doi:10.1021/ie50167a018.
- Jump up^ O.E.D. 2nd Edition 2005
- Jump up^ Müller, Werner E. G. (2003). "The Origin of Metazoan Complexity: Porifera as Integrated Animals". Integrative and Comparative Biology 43 (1): 3–10. doi:10.1093/icb/43.1.3.
- Jump up^ Sabiston textbook of surgery board review, 7th edition. Chapter 5 wound healing, question 14
- Jump up^ Schauss, A., Stenehjem, J., Park, J., Endres, J., and Clewell, A. (2012). "Effect of the novel low molecular weight hydrolyzed chicken sternal cartilage extract, BioCell Collagen, on improving osteoarthritis-related symptoms: A randomized, double-blind, placebo-controlled trial". Journal of Agricultural and Food Chemistry 60 (16): 4096–101.doi:10.1021/jf205295u. PMID 22486722.
- ^ Jump up to:a b Dermal Fillers | The Ageing Skin. Pharmaxchange.info. Retrieved on 2013-04-21.
- Jump up^ Cunniffe, G; F O'Brien (2011). "Collagen scaffolds for orthopedic regenerative medicine".The Journal of the Minerals, Metals and Materials Society 63 (4): 66–73.Bibcode:2011JOM....63d..66C. doi:10.1007/s11837-011-0061-y.
- Jump up^ Oliveira, S; R Ringshia; R Legeros; E Clark; L Terracio; C Teixeira M Yost (2009). "An improved collagen scaffold for skeletal regeneration". Journal of Biomedical Materials 94(2): 371–379. doi:10.1002/jbm.a.32694. PMC 2891373. PMID 20186736.
- Jump up^ Blow, Nathan (2009). "Cell culture: building a better matrix". Nature Methods 6 (8): 619–622. doi:10.1038/nmeth0809-619.
- Jump up^ Birbrair, Alexander; Zhang, Tan; Files, Daniel C.; Mannava, Sandeep; Smith, Thomas; Wang, Zhong-Min; Messi, Maria L.; Mintz, Akiva; Delbono, Osvaldo (2014-11-06). "Type-1 pericytes accumulate after tissue injury and produce collagen in an organ-dependent manner". Stem Cell Research & Therapy 5 (6): 122. doi:10.1186/scrt512. ISSN 1757-6512. PMC 4445991. PMID 25376879.
- Jump up^ Singh, O; SS Gupta; M Soni; S Moses; S Shukla; RK Mathur (2011). "Collagen dressing versus conventional dressings in burn and chronic wounds: a retrospective study".Journal of Cutaneous and Aesthetic Surgery 4 (1): 12–16. doi:10.4103/0974-2077.79180. PMC 3081477. PMID 21572675.
- ^ Jump up to:a b c d e f Szpak, Paul (2011). "Fish bone chemistry and ultrastructure: implications for taphonomy and stable isotope analysis". Journal of Archaeological Science 38 (12): 3358–3372. doi:10.1016/j.jas.2011.07.022.
- Jump up^ Diegelmann, Robert. "Collagen Metabolism". Medscape.com. Medscape. Retrieved4 December 2014.
- Jump up^ Gorres, K. L.; Raines, R. T. (2010). "Prolyl 4-hydroxylase". Crit. Rev. Biochem. Mol. Biol. 45 (2): 106–24. doi:10.3109/10409231003627991. PMC 2841224.PMID 20199358.
- Jump up^ Myllylä, R.; Majamaa, K.; Günzler, V.; Hanauske-Abel, H. M.; Kivirikko, K. I. (1984). "Ascorbate is consumed stoichiometrically in the uncoupled reactions catalyzed by propyl 4-hydroxylase and lysyl hydroxylase". J. Biol. Chem. 259 (9): 5403–5. PMID 6325436.
- Jump up^ Houck, J. C.; Sharma, V. K.; Patel, Y. M.; Gladner, J. A. (1968). "Induction of Collagenolytic and Proteolytic Activities by AntiInflammatory Drugs in the Skin and Fibroblasts". Biochemical Pharmacology 17 (10): 2081–2090. doi:10.1016/0006-2952(68)90182-2. PMID 4301453.
- Jump up^ Al-Hadithy, H.; Isenberg, DA; et al. (1982). "Neutrophil function in systemic lupus erythematosus and other collagen diseases". Ann Rheum Dis 41 (1): 33–38.doi:10.1136/ard.41.1.33. PMC 1000860. PMID 7065727.
- Jump up^ Hulmes, D. J. (2002). "Building collagen molecules, fibrils, and suprafibrillar structures". J Struct Biol 137 (1–2): 2–10. doi:10.1006/jsbi.2002.4450. PMID 12064927.
- ^ Jump up to:a b Hulmes, D. J. (1992). "The collagen superfamily—diverse structures and assemblies".Essays Biochem 27: 49–67. PMID 1425603.
- ^ Jump up to:a b c Orgel, J. P.; Irving, TC; et al. (2006). "Microfibrillar structure of type I collagen in situ". PNAS 103 (24): 9001–9005. Bibcode:2006PNAS..103.9001O.doi:10.1073/pnas.0502718103. PMC 1473175. PMID 16751282.
- ^ Jump up to:a b Hulmes, D. J. & Miller, A. (1979). "Quasi-hexagonal molecular packing in collagen fibrils". Nature 282 (5741): 878–880. Bibcode:1979Natur.282..878H.doi:10.1038/282878a0. PMID 514368.
- Jump up^ Perumal, S.; Antipova, O. & Orgel, J. P. (2008). "Collagen fibril architecture, domain organization, and triple-helical conformation govern its proteolysis". PNAS 105 (8): 2824–2829. Bibcode:2008PNAS..105.2824P. doi:10.1073/pnas.0710588105.PMC 2268544. PMID 18287018.
- Jump up^ Sweeney, S. M.; Orgel, JP; et al. (2008). "Candidate Cell and Matrix Interaction Domains on the Collagen Fibril, the Predominant Protein of Vertebrates". J Biol Chem 283 (30): 21187–21197. doi:10.1074/jbc.M709319200. PMC 2475701. PMID 18487200.
- Jump up^ Twardowski, T.; Fertala, A.; et al. (2007). "Type I collagen and collagen mimetics as angiogenesis promoting superpolymers". Curr Pharm Des 13 (35): 3608–3621.doi:10.2174/138161207782794176.
- Jump up^ Minary-Jolandan, M; Yu, MF (2009). "Nanomechanical heterogeneity in the gap and overlap regions of type I collagen fibrils with implications for bone heterogeneity".Biomacromolecules 10 (9): 2565–70. doi:10.1021/bm900519v. PMID 19694448.
- Jump up^ Ross, M. H. and Pawlina, W. (2011) Histology, 6th ed., Lippincott Williams & Wilkins, p. 218.
- Jump up^ Söderhäll, C.; Marenholz, I.; Kerscher, T.; Rüschendorf, F; Rüschendorf, F.; Esparza-Gordillo, J.; Mayr, G; et al. (2007). "Variants in a Novel Epidermal Collagen Gene (COL29A1) Are Associated with Atopic Dermatitis". PLoS Biology 5 (9): e242.doi:10.1371/journal.pbio.0050242. PMC 1971127. PMID 17850181.
- ^ Jump up to:a b Mahajan, VB, Olney, AH, Garrett, P, Chary, A, Dragan, E, Lerner, G, Murray, J & Bassuk, AG; Olney; Garrett; Chary; Dragan; Lerner; Murray; Bassuk (2010). "Collagen XVIII mutation in Knobloch syndrome with acute lymphoblastic leukemia". American journal of medical genetics. Part A 152A (11): 2875–9. doi:10.1002/ajmg.a.33621.PMC 2965270. PMID 20799329.
- Jump up^ Gajko-Galicka, A (2002). "Mutations in type I collagen genes resulting in osteogenesis imperfecta in humans" (PDF). Acta biochimica Polonica 49 (2): 433–41.PMID 12362985.
- Jump up^ Horton, WA, Campbell, D, Machado, MA & Chou, J; Campbell; Machado; Chou (1989). "Type II collagen screening in the human chondrodysplasias". American Journal of Medical Genetics 34 (4): 579–83. doi:10.1002/ajmg.1320340425. PMID 2624272.
- Jump up^ Hamel, BCJ; Pals, G.; Engels, CHAM; Akker, E.; Boers, GHJ; van Dongen, PWJ; Steijlen, PM (28 June 2008). "Ehlers-Danlos syndrome and type III collagen abnormalities: a variable clinical spectrum". Clinical Genetics 53 (6): 440–446. doi:10.1111/j.1399-0004.1998.tb02592.x. PMID 9712532.
- Jump up^ Kashtan, CE (1993) "Collagen IV-Related Nephropathies (Alport Syndrome and Thin Basement Membrane Nephropathy)", in RA Pagon, TD Bird, CR Dolan, K Stephens & MP Adam (eds), GeneReviews, University of Washington, Seattle, Seattle WA PMID 20301386.
- Jump up^ Shuster, S (2005). "Osteoporosis, a unitary hypothesis of collagen loss in skin and bone".Medical Hypotheses 65 (3): 426–32. doi:10.1016/j.mehy.2005.04.027.PMID 15951132.
- Jump up^ Fratzl, P. (2008). Collagen: Structure and Mechanics. New York: Springer. ISBN 0-387-73905-X.
- Jump up^ Buehler, M. J. (2006). "Nature designs tough collagen: Explaining the nanostructure of collagen fibrils". PNAS 103 (33): 12285–12290. Bibcode:2006PNAS..10312285B.doi:10.1073/pnas.0603216103. PMC 1567872. PMID 16895989.
- Jump up^ Zylberberg, L.; Laurin, M. (2011). "Analysis of fossil bone organic matrix by transmission electron microscopy". Comptes rendus Palevol 11 (5–6): 357–366.doi:10.1016/j.crpv.2011.04.004.
- Jump up^ "Gelatin's Advantages: Health, Nutrition and Safety". gmap-gelatin.com. Archived fromthe original on May 5, 2006.
- Jump up^ European Food Safety Authority - EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific Opinion on the substantiation of a health claim related to collagen hydrolysate and maintenance of joints pursuant to Article 13(5) of Regulation (EC) No 1924/20061. EFSA Journal 2011;9(7):2291.
- Jump up^ Walker, Amélie A. (May 21, 1998). "Oldest Glue Discovered". Archaeology.
- Jump up^ Ennker, I. C.; Ennker, JüRgen; et al. (1994). "Formaldehyde-free collagen glue in experimental lung gluing". Ann Thorac Surg. 57 (6): 1622–1627. doi:10.1016/0003-4975(94)90136-8. PMID 8010812.
- Jump up^ Wyckoff, R.; Corey, R. & Biscoe, J. (1935). "X-ray reflections of long spacing from tendon". Science 82 (2121): 175–176. Bibcode:1935Sci....82..175W.doi:10.1126/science.82.2121.175. PMID 17810172.
- Jump up^ Clark, G.; Parker, E.; Schaad, J. & Warren, W. J. (1935). "New measurements of previously unknown large interplanar spacings in natural materials". J. Amer. Chem. Soc 57(8): 1509. doi:10.1021/ja01311a504.
- Jump up^ Balasubramanian, D . (October 2001). "GNR — A Tribute". Resonance (Indian Academy of Sciences) 6 (10).
- Jump up^ Leonidas, Demetres D.; Chavali, GB; et al. (2001). "Binding of Phosphate and pyrophosphate ions at the active site of human angiogenin as revealed by X-ray crystallography". Protein Science 10 (8): 1669–1676. doi:10.1110/ps.13601.PMC 2374093. PMID 11468363.
- Jump up^ Subramanian, Easwara (2001). "Obituary: G.N. Ramachandran". Nature Structural & Molecular Biology 8 (6): 489–491. doi:10.1038/88544. PMID 11373614.
- ^ Jump up to:a b c Saad, Mohamed (Oct 1994). Low resolution structure and packing investigations of collagen crystalline domains in tendon using Synchrotron Radiation X-rays, Structure factors determination, evaluation of Isomorphous Replacement methods and other modeling. PhD Thesis, Université Joseph Fourier Grenoble I. pp. 1–221.doi:10.13140/2.1.4776.7844.
- Jump up^ Fraser, R. D.; MacRae, T. P. & Suzuki, E. (1979). "Chain conformation in the collagen molecule". J Mol Biol 129 (3): 463–481. doi:10.1016/0022-2836(79)90507-2.PMID 458854.
- Jump up^ Okuyama, K.; Okuyama, K; et al. (1981). "Crystal and molecular structure of a collagen-like polypeptide (Pro-Pro-Gly)10". J Mol Biol 152 (2): 427–443. doi:10.1016/0022-2836(81)90252-7. PMID 7328660.
- Jump up^ Traub, W.; Yonath, A. & Segal, D. M. (1969). "On the molecular structure of collagen".Nature 221 (5184): 914–917. Bibcode:1969Natur.221..914T. doi:10.1038/221914a0.
- Jump up^ Bella, J.; Eaton, M.; Brodsky, B.; Berman, H. M. (1994). "Crystal and molecular structure of a collagen-like peptide at 1.9 A resolution". Science 266 (5182): 75–81.Bibcode:1994Sci...266...75B. doi:10.1126/science.7695699. PMID 7695699.
- Jump up^ Jesior, J. C.; Miller, A. & Berthet-Colominas, C. (1980). "Crystalline three-dimensional packing is general characteristic of type I collagen fibrils". FEBS Lett 113 (2): 238–240.doi:10.1016/0014-5793(80)80600-4. PMID 7389896.
- Jump up^ Fraser, R. D. B. & MacRae, T. P. (1981). "Unit cell and molecular connectivity in tendon collagen". Int. J. Biol. Macromol. 3 (3): 193–200. doi:10.1016/0141-8130(81)90063-5.
- Jump up^ Fraser, R. D.; MacRae, T. P.; Miller, A. (1987). "Molecular packing in type I collagen fibrils". J Mol Biol 193 (1): 115–125. doi:10.1016/0022-2836(87)90631-0.PMID 3586015.
- Jump up^ Wess, T. J.; Hammersley, AP; et al. (1998). "Molecular packing of type I collagen in tendon". J Mol Biol 275 (2): 255–267. doi:10.1006/jmbi.1997.1449. PMID 9466908.
- Jump up^ Raspanti, M.; Ottani, V.; Ruggeri, A. (1990). "Subfibrillar architecture and functional properties of collagen: a comparative study in rat tendons". J Anat. 172: 157–164.PMC 1257211. PMID 2272900.
- Jump up^ Holmes, D. F.; Gilpin, C. J.; Baldock, C.; Ziese, U.; Koster, A. J.; Kadler, K. E. (2001)."Corneal collagen fibril structure in three dimensions: Structural insights into fibril assembly, mechanical properties, and tissue organization". PNAS 98 (13): 7307–7312.Bibcode:2001PNAS...98.7307H. doi:10.1073/pnas.111150598. PMC 34664.PMID 11390960.
- Jump up^ Holmes, D. F.; Kadler, KE (2006). "The 10+4 microfibril structure of thin cartilage fibrils". PNAS 103 (46): 17249–17254. Bibcode:2006PNAS..10317249H.doi:10.1073/pnas.0608417103. PMC 1859918. PMID 17088555.
- Jump up^ Okuyama, K; Bächinger, HP; Mizuno, K; Boudko, SP; Engel, J; Berisio, R; Vitagliano, L (2009). "Comment on Microfibrillar structure of type I collagen in situ by Orgel et al. (2006), Proc. Natl Acad. Sci. USA, 103, 9001–9005". Acta Crystallogr D Biol Crystallogr 65 (Pt9): 1009–10. doi:10.1107/S0907444909023051. PMID 19690380.
- Jump up^ Narayanaswamy, Radhakrishnan; Shanmugasamy, Sangeetha; Shanmugasamy, Sangeetha; Gopal, Ramesh; Mandal, Asit (2011). "Bioinformatics in crosslinking chemistry of collagen with selective crosslinkers". BMC Research Notes 4: 399. doi:10.1186/1756-0500-4-399.
External links
12 types of collagen
- Database of type I and type III collagen mutations
- Science.dirbix Collagen
- Collagen Stability Calculator
- Computer-generated animations of the assembly of Type I and Type IV Collagens
- Integrin-Collagen interface, PMAP (The Proteolysis Map)—animation
- Integrin-Collagen binding model, PMAP (The Proteolysis Map)—animation
- Collagen-Integrin atomic detail, PMAP (The Proteolysis Map)—animation
- Saad M. (1994) "Low resolution structure and packing investigations of collagen crystalline domains in tendon using Synchrotron Radiation X-rays, Structure factors determination, evaluation of Isomorphous Replacement methods and other modeling." PhD Thesis, Université Joseph Fourier Grenoble 1
////////