BIA 10-2474
cas 1233855-46-3
3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide
1
H-Imidazole-1-carboxamide,
N-cyclohexyl-
N-methyl-4-(1-oxido-3-pyridinyl)-
C
16 H
20 N
4 O
2, 300.36
BIA 10-2474 is an experimental
fatty acid amide hydrolase inhibitor
[1] developed by the Portuguese pharmaceutical company
Bial-Portela & Ca. SA. The drug was developed to relieve pain,
[2][3] to ease mood and anxiety problems, and to improve movement coordination linked to neurodegenerative illnesses.
[4] It interacts with the human
endocannabinoid system.
[5][6] It has been linked to
severe adverse events affecting 5 patients in a drug trial in Rennes, France, and at least one death, in January 2016.
[7]

Synthesis
BIAL – PORTELA & Cª, S.A.
Example 5. 3-(l-(cyclohexyl(methyl)carbamoyl-lfl-imidazol-4-yl)pyridine l-oxide (compound A)
C
16H
20N
4O C16H20N4O2
MW 284,36 MW 300,36
To a solution of
N-cyclohexyl-N-methyl-4-(pyridm-3-yl)-lH-imidazole-l-carboxamide in
dichioromethane at 25°C was added peracetic acid (38%; the concentration
is not critical, and may be varied) in a single portion. The reaction
mixture was then maintained at 25°C for at least 20 h, whereupon the
reaction was washed four times with water (in some embodiments, the
water for the extraction step may be supplemented with a small amount
(e.g. 1%) of acetic acid, which helps to promote product solubility in
the DCM). The dichioromethane solution was then filtered prior to
diluting with 2-propanol. Dichioromethane (50%) was then distilled off
under atmospheric pressure, whereupon, 2-propanol was charged at the
same rate as the distillate was collected. The distillation was
continued until >90% of the dichioromethane was collected. The
resulting suspension was then cooled to 20°C and aged for at least 30
min. prior to cooling to 0°C and aging for a further 60 min. The
reaction mixture was then filtered and the product washed with
additional 2-propanol, before drying at 50°C under vacuum to afford the
title compound as an off-white crystalline solid.
The purity of the product was ascertained by HPLC, with identity
confirmable by NMR. The yield was consistently >80% in several
production runs.
PATENT
WO 2012015324
Example 1. Preparation of N-cyclohexyl-N-methyl-4-(pyridin-3yl)-lH-imidazole-l-carboxamide
C
8H
7N
3 C
15H
1 1N
30
2 C
16H
20N
4O
MW 145,16 MW 265,27 MW 284,36
To a suspension of 3-(l/ -imidazol-4-yl)pyridine in tetrahydrofuran
(THF) containing pyridine at 25°C was slowly added a solution of phenyl
chloroformate in THF over 60 to 90 min. The resulting fine white
suspension was then maintained at 25°C for at least 60 min. before the
addition of N-methyl- -cyclohexylamine in a single portion, causing the
suspension to thin and become yellow in colour. The reaction mixture was
then stirred for 90 min. before filtering and washing the filter cake
with additional THF. The mother liquors were then maintained at 25°C for
at least 18 h, whereupon 65% of the volume of THF was distilled off
under atmospheric pressure. The resulting solution was then diluted with
2-propanol and maintained at > 50°C for 10 min. prior to cooling
down to 20°C. The resulting suspension was aged at 20°C for 15 min.
prior to cooling to 0°C and aging for a further 60 min. The reaction
mixture was then filtered and the product was washed with additional
2-propanol, before drying at 50°C under vacuum to afford the title
compound as an off-white crystalline solid.
The purity of the product was ascertained by HPLC, with identity
confirmable by NMR. The yield was consistently around 50% in several
production runs.
Example 2. 3-(l-(cyclohexyl(methyl)carbamoyl-l//-imidazol-4-yl)pyridine 1 -oxide (compound A)
C
16H
20N
4O Ci6H2oN
40
2
MW 284,36 MW 300,36
To a solution of
N-cyclohexyl-N-methyl-4-(pyridin-3-yl)-lH-imidazole-l-carboxamide in
dichloromethane at 25°C was added peracetic acid (38%; the concentration
is not critical, and may be varied) in a single portion. The reaction
mixture was then maintained at 25°C for at least 20 h, whereupon the
reaction was washed four times with water. The dichloromethane solution
was then filtered prior to diluting with 2-propanol. Dichloromethane
(50%) was then distilled off under atmospheric pressure, whereupon,
2-propanol was charged at the same rate as the distillate was collected.
The distillation was continued until >90% of the dichloromethane was
collected. The resulting suspension was then cooled to 20°C and aged
for at least 30 min. prior to cooling to 0°C and aging for a further 60
min. The reaction mixture was then filtered and the product washed with
additional 2-propanol, before drying at 50°C under vacuum to afford the
title compound as an off-white crystalline solid.
The purity of the product was ascertained by HPLC, with identity
confirmable by NMR. The yield was consistently >80% in several
production runs. It will be appreciated that this gives an overall yield
of compound A many times greater than that achieved in the prior art.
In a further run of this synthesis, in a 2L reactor to a mixture of
N-cyclohexyl-N-methyl-4-(pyridin-3-yl)-l H- imidazole-l-carboxamide (90
g, 317 mmol) and dichloromethane (1350 ml) was added peracetic acid (84
ml, 475 mmol). The reaction mixture was stirred at 25°C. Completion of
the reaction was monitored by HPLC for the disappearance of
N-cyclohexyl-N-methyl-4-(pyridin-3-yl)-lH- imidazole- 1-carboxamide.
After reaction completion a solution of sodium metabisulfite (60.2 g,
317 mmol) in water (270ml) was added to the reaction mixture maintaining
the temperature below 30°C. After phase separation the organic phase
was washed with water. After phase separation the organic phase was
concentrated at atmospheric pressure until 5 vol. Then solvent was
swapped to isopropanol (1350 ml) and the suspension was cooled to 0°C
during 4 hours and stirred at that temperature for 1 hour. The resulting
solid was collected by filtration and was rinsed with water (270 ml)
and isopropanol (270 ml) to afford a white crystalline solid in 84.8g
(89%).
PATENT
WO 2010074588
Preparation of compound 362 a) N-cyclohexyl-N-methyl-4-(pyridin-3-yl)- 1 H-imidazole- 1 -carboxamide
To a stirred suspension of 3-( 1 H-imidazol-4-yl)pyridine
dihydrochloride (1.745 g, 8 mmol) in a mixture of tetrahydrofuran (29
mL) and DMF (2.90 mL) was added potassium 2-methylpropan-2-olate (1.795
g, 16.0 mmol) and the mixture was refluxed for 30 minutes. The resulting
brown suspension was cooled to room temperature and treated with
pyridine (0.979 mL, 12 mmol) and N,N-dimethylpyridin-4-amine (0.098 g,
0.8 mmol), followed by the addition of cyclohexyl(methyl)carbamic
chloride (1.476 g, 8.4 mmol). The reaction was heated to 90
0C
overnight, whereupon the mixture was diluted with water and extracted
with ethyl acetate. The organic phase was dried (MgSO^) and filtered.
After evaporation, the crude product was chromatographed over silica gel
using a dichloromethane/methanol (9:1) mixture. Homogenous fractions
were pooled and evaporated to leave a white powder, (160 mg, 7 %).
b) 3-( 1 -(cyclohexyl(methyl)carbamoyl)- 1 H-imidazol-4-yl)pyridine 1 -oxide
To a stirred solution of N-cyclohexyl-N-methyl-4-(pyridin-3-yl)-l
H-imidazole- 1 -carboxamide (90 mg, 0.317 mmol) in chloroform (5 mL) was
added 3-chlorobenzoρeroxoic acid (149 mg, 0.475 mmol) in one portion.
The reaction was allowed to stir at room temperature for 20 h. TLC
showed the reaction to be complete and the mixture was evaporated to
dryness. The residue was triturated with ether and the resulting white
crystals were filtered off and dried in air. Recrystallisation from hot
isopropanol gave a white powder (46 mg, 46 %).
Structure and action
French newspaper
Le Figaro
has obtained Bial study protocol documents listing the the chemical
name of BIA-10-2474 as
3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide.
[8] A Bial news release described BIA-10-2474 as “a long-acting inhibitor of FAAH”.
[9]
Fatty acid amide hydrolase (FAAH) is an enzyme which degrades
endocannabinoid neurotransmitters like
anandamide,
[10] which relieves pain and can affect eating and sleep patterns.
[11][12] FAAH inhibitors have been proposed for a range of nervous-system disorders including anxiety, alcoholism, pain and nausea.
The Portuguese pharmaceutical company
Bial holds several patents on FAAH enzyme inhibitors.
[12][13][14][15]
No details of the preclinical testing of this molecule have been made
public by the manufacturer Bial. However, the French newspaper
Le Figaro has obtained and published an apparently legitimate copy of the full
clinical trial protocol (BIA-102474-101).
[8] The protocol presents a summary of what appears to be a full package of
pharmacodynamic,
pharmacokinetic and
toxicological studies that might be expected to support a
first-in-man study, including
safety pharmacology
studies in two species (rat, dog) and repeated dose toxicity studies in
four species (13 week sub-chronic studies in mouse, rat, dog and
monkey). The summary presented however includes no assessment of the
relevance of the animal species selected for study (that is, in terms of
physiological and genetic similarities with humans and the
mechanism of action of the study drug).
Of note, few adverse events were observed in any of the studies, with the 13-week oral
No Observed Adverse Effect Level
(NOAEL) varying between 10 mg/kg/day in mice to 75 mg/kg/day in
monkeys. The authors suggest that these were the maximum doses tested in
these studies, though it is not clear. The authors also report no
effects of significance in the animal models used for the
CNS safety pharmacology studies, which studied a dose of up to 300 mg/kg/day.
[8]
Notably absent from the protocol are calculations of receptor
occupancy; predictions of in vivo ligand binding saturation levels;
measures of
target affinity;
or assessment of the molecule’s activity in non-target tissues or
non-target binding interactions as suggested by the European guidance
for Phase I studies,
[16] assuming BIA 10-2474 could be considered ‘high risk’).
[8]
The trial protocol makes no reference to chimpanzee studies (only
monkeys) which contradicts a previous statement to the media in which
the French Health Minister stated that the drug had been tested on
animals including
chimpanzees.
[4][17] [18] Some experts had remarked that
drug testing in chimpanzees was unlikely.
[19]
These findings provide no explanation for the type and severity of
events observed in Rennes. In describing the rationale for the starting
dose, the authors conclude that:
No target organ was identified during toxicology studies and few adverse clinical findings were observed at the highest dose tested. For the single ascending dose part [of the clinical trial], a starting dose of 0.25 mg was judged to be safe for a first-in-human administration. [8]
The protocol defines no starting dose for the multi-dose treatment
groups, noting that this will be based on the outcome of the single dose
portion of the trial (an approach known as
adaptive trial design). The authors note that nonetheless, the starting dose will not exceed 33% of the
maximum tolerated dose (MTD) identified in the single dose groups (or 33% of the maximum administered dose if the MTD is not reached).
[8]
Death and serious adverse events during phase I clinical trial
In July 2015 Biotrial, a
contract research organization, began testing the drug in a human
phase one clinical trial for the manufacturer. The study was approved by French regulatory authority, the
Agence Nationale de Sécurité du Médicament (ANSM), on June 26, 2015, and by the Brest regional ethics committee on July 3, 2015.
[20] The trial commenced on July 9, 2015,
[21] in the city of
Rennes,
and recruited 128 healthy volunteers, both men and women aged 18 to 55.
According to French authorities, the study employed a three-stage
design with 90 of the volunteers having received the drug during the
first two stages of the trial, with no
serious adverse events being reported .
[17][20]
Participants of the study were to receive €1,900 and, in turn, asked to
stay at Biotrial’s facility for two weeks during which time they would
take the drug for ten days and undergo tests.
[22]
In the third stage of the trial evaluating multiple doses, six male
volunteers received doses by mouth, starting on 7 January 2016. The
first volunteer was hospitalized at the
Rennes University Hospital on January 10, became brain dead,
[17][23][24][25] and died on January 17.
[26] The other five men in the same dosage group were also hospitalized, in the period of January 10 through January 13
[27] four of them suffering injuries including deep hemorrhagic and necrotic lesions seen on
brain MRI.
[7] The six men who were hospitalised were the group which received the highest dose.
[26]
A neurologist at the University of Rennes Hospital Center, Professor
Pierre-Gilles Edan, stated in a press conference with the French
Minister for Health, that 3 of the 4 men who were displaying
neurological symptoms “already have a severe enough clinical picture to
fear that even in the best situation there will be an irreversible
handicap” and were being given corticosteroids to control the
inflammation.
[27] The sixth man from the group was not showing adverse effects but had been hospitalized for observation.
[25][28][29] Biotrial stopped the experiment on January 11, 2016.
[4]
No details of the trial have been made public by the manufacturer Bial. The study does not appear in searches of any of the key
clinical trial registries, including
EudraCT and
ClinicalTrials.gov which would normally contain details of approved clinical studies.
[30][31][32][33]
The trial protocol published by Le Figaro provides extensive detail on
what was planned for the study, but many details of the key multi-dose
part are not included and were to have been finalised at the conclusion
of the single-dose part of the trial.
[8]
The French health minister
Marisol Touraine called the event “an accident of exceptional gravity” and promised to investigate the matter.
[4] On January 18 it was reported authorities were investigating if a manufacturing or transport error might be involved.
[34]
Le Figaro posted
a 96-page clinical study protocol for BIA 10-2474 that the French newspaper procured from an unnamed source.
According to the document, BIA 10-2474 is 3-(1-(cyclohexyl(methyl)carbamoyl)-1H-imidazol-4-yl)pyridine 1-oxide.
BIA 10-2474 “is designed to act as a long-active and reversible
inhibitor of brain and peripheral FAAH,” notes the protocol. The
compound “increases anandamide levels in the central nervous system and
in peripheral tissues.”
The clinical trial protocol also notes that the company tested BIA
10-2474 on mice, rats, dogs, and monkeys for effects on the heart,
kidneys, and gastrointestinal tract, among other pharmacological and
toxicological evaluations.
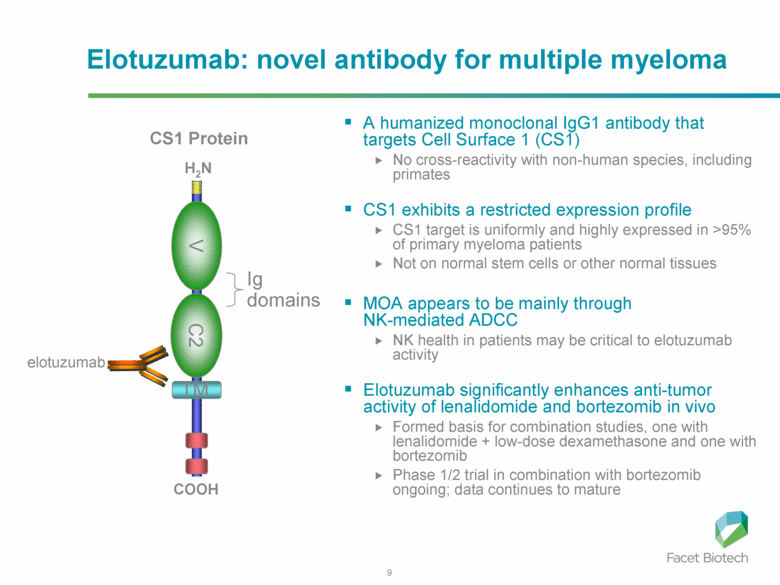
Six men in a Phase I clinical trial were admitted to
the University Hospital Center of Rennes, France, (shown here) because
of adverse reactions.Six men in a Phase I clinical trial were admitted
to the University Hospital Center of Rennes, France, (shown here)
because of adverse reactions.
Credit: Mathieu Pattier/SIPA/Newscom
One man is dead and five men were hospitalized after participating in a Phase I clinical trial in Rennes, France
The clinical trial,
conducted by the company Biotrial on behalf of the Portuguese pharmaceutical firm Bial,
was evaluating a pain relief drug candidate called BIA 10-2474 that
inhibits fatty acid amide hydrolase (FAAH) enzymes. Blocking these
enzymes prevents them from breaking down cannabinoids in the brain, a
family of compounds that includes the euphoria-inducing neurotransmitter
anandamide and Δ
9-tetrahydrocannabinol, the major psychoactive component of marijuana.
Phase I clinical trials are conducted to check a drug candidate’s
safety profile in healthy, paid volunteers. In this case, the drug
caused hemorrhagic and necrotic brain lesions in five out of six men in a
group who received the highest doses of the drug, said Gilles Edan, a
neurologist at the
University Hospital Center of Rennes.

The most severely affected man was pronounced brain-dead after
hospitalization and then died on Jan. 17. Four men remain in the
hospital in stable condition. The only man in the high-dose group who
had no adverse symptoms has been released from the hospital.
Clinical trials are an essential part of the drug development
process. In order to get life-improving and life-saving medicines to
patients, they first have to go through an extensive series of tests.
Even before a drug makes it to Phase 1 testing, where its safety, dosage
amount, and side effects are tested in a small group of humans, it will
undergo testing in animals. As a result, it is not common for a
medicine undergoing clinical tests to have a very serious adverse effect
on a human. This makes you wonder what happened to a group of patients
involved in a clinical study in Rennes, France.
According to
news reports,
a drug undergoing testing in a French clinic has left one person dead,
two others with what may be permanent brain damage, and and two others
critically ill. The drug has thus far been unnamed, but it appears to
have been produced by the Portuguese company Bial. The French health
minister has stated the drug acted on natural receptors found in the
body known as endocannibinoids, which regulate mood and appetite. It did
not contain cannabis or anything derived from it, as was originally
reported. All six trial participants were administered the doses
simultaneously.
The trial was being performed at Biotrial, a French-based firm that
was formed in 1989 and has conducted thousands of trials. A message on
the company’s website stated that they are working with health
authorities to understand the cause of the accident, while extending
thoughts to the patients and their families. Bial has disclosed the drug
was a FAAH (fatty acid amide hydrolase) inhibitor, which is an enzyme
produced in the brain and elsewhere that breaks down neurotransmitters
called endocannabinoids. Two scientists from the Nottingham Medical
School who have worked with FAAH tried over the weekend to try and
identify the drug by examining a list of drugs Bial currently has in its
pipeline. They believe the culprit is one identified by the codename
BIA 10-2474. That same codename appeared on a recruitment form that was
given to a volunteer, which was published in a French newspaper. Little
more is known about it, and there does not appear to be any entry for it
in clinical trial registries.
The French health ministry is reporting the six patients were all in
good health prior to taking the oral medicine, which was administered to
90 volunteers. The trial recruited 128 individuals, and the remaining
participants received a placebo. Health minister Marisol Touraine,
describing the situation as a very serious accident, noted the patients
were taking part in a trial in Brittany, Rennes involving a medicine
developed by a “European laboratory”, refusing to comment further until
additional information became available. She has also asked the
Inspector General of Social Affairs to lead an investigation into the
circumstances around the trial, which has obviously been suspended. She
notes the drug had been tested on animals, including chimpanzees.
France’s National Agency for Medicine and Health Products Safety
approved the trial on in June 2015.
One thing we do know is that the trial was a Phase 1 clinical study that included 90 healthy volunteers.
Regulations
that oversee all clinical trials in Europe do attempt to minimize the
risk associated with trials, but there is always a risk involved with
administering an unapproved medicine to humans. At this time the chief
neuroscientist at the hospital where the patients are being treated has
said there is no known antidote for the drug.
The drug, administered to men between the ages of 28 and 49, was
intended to treat mood disorders such as anxiety. While the men were
administered varying doses, the patients who are hospitalized were
taking the drug “regularly”.

Old 2006 case
While safety issues like this are rare, they are not unheard of.
In 2006, a clinical trial in London
left six men ill. All were taking part in a study testing a drug
designed to fight auto-immune disease and leukemia. Within hours of
taking the drug TGN1412, all experienced a serious reaction, were
admitted to intensive care, and had to be treated for organ failure. Two
became critically ill, with one eventually losing all of his fingers
and toes. All were told they would have a higher risk of developing
cancers or auto-immune diseases.
This of course led many to wonder about the future of trials, and whether the situation could happen again. The
Duff Report,
written in response to the TGN1412 trial, noted the medicine should
have been tested in one person at a time. It also helped to put
additional safety measures in place. The Medicines and Health Products
Regulatory Agency (MHRA) now requires committees to look at pre-clinical
data to determine the proper initial dose, and rules are in place to
stop the trial if unintended reactions occur.
However, since patients can fall ill immediately after being administered a medication, certain risks will still exist.
The company that manufactured TGN1412, TeGenero Immuno Therapeutics,
later went bankrupt. However the drug was later purchased by a Russian
investor and renamed TABO8. TheraMAB, a Russian biotech company, then
conducted a new trial of the drug in a much lower dose. A later Phase 2
study was started in patients with Rheumatoid Arthritis.
Other pharmaceutical companies, including
Merck,
Pfizer,
Johnson & Johnson,
Sanofi and
Vernalis, have previously taken other FAAH inhibitors into clinical trials without experiencing such adverse events (e.g. respectively,
MK-4409,
[35][36] PF-04457845,
JNJ-42165279,
[37] SSR411298 and V158866.
[38][39] Related enzyme inhibitor compounds such as
URB-597 and
LY-2183240 have been sold illicitly as
designer drugs,
[40][41]
all without reports of this type of toxicity emerging, so the mechanism
of the toxicity observed with BIA 10-2474 remains poorly understood.
Following the events in Rennes, Janssen announced that it was
temporarily suspending dosing in two Phase II clinical trials with its
own FAAH inhibitor
JNJ-42165279,
headlining the decision as “precautionary measure follows safety issue
with different drug in class”. Janssen was emphatic that no serious
adverse events had been reported in any of the clinical trials with
JNJ-42165279 to date. The suspension is to remain in effect until more
information is available about the BIA 10-2474 study.
[42]
References
- 1 “BRIEF-Bial says firmly committed to ensure wellbeing of test participants”. Reuters. 15 January 2016.
- 2 “BIA 10-2474”. Biocentury.com. Retrieved 2016-01-17.
- 3 “BIA 102474”. Adisinsight/springer.com. Retrieved 2016-01-17.
- 4 Adamson B (January 15, 2016). “Botched Drug Trial Leaves 1 Brain Dead, 5 in Hospital”. ABC, AP. Retrieved January 16, 2016.
- 5 Angeline Benoit,Makiko Kitamura (15 January 2016). “France Ties Brain-Dead Person to Tests of Bial-Portela Drug”. Bloomberg.com.
- 6 “France/Monde – Essai thérapeutique : 90 personnes ont pris la molécule”. Ledauphine.com. Retrieved 2016-01-17.
- 7 Enserink, Martin (2016). “More Details Emerge on Fateful French Drug Trial” (online). Science (January 16). Retrieved 16 January 2016.
- 8 “Drame de Rennes : le protocole de l’essai clinique en accusation”. sante.lefigaro.fr. Retrieved 2016-01-21.
- 9 “News Release – Phase I Clinical Trial Rennes”. http://www.bial.com. Retrieved 2016-01-21.
- 10 Debora Mackenzie. “Six in hospital after French pain relief drug trial goes wrong”. New Scientist.
- 11 “The Discovery and Development of Inhibitors of Fatty Acid Amide Hydrolase (FAAH)”. PubMed Central.
- 12 “Patent WO2015012708A1 – Imidazolecarboxamides and their use as faah inhibitors – Google Patents”. Google.com. Retrieved 2016-01-17.
- 13 “Patent WO2015016729A1 – Urea compounds and their use as faah enzyme inhibitors”. Google.com. Retrieved 2016-01-17.
- 14 “PT2014000052 UREA COMPOUNDS AND THEIR USE AS FAAH ENZYME INHIBITORS”. Patentscope.wipo.int. Retrieved 2016-01-17.
- 15 WO application 2015016729,
Laszlo Erno KISS, Rita GUSMÃO DE NORONHA, Carla Patrícia ROSA DA COSTA
PEREIRA, Rui PINTO, “Urea compounds and their use as faah enzyme
inhibitors”, published Feb 5, 2015, assigned to BIAL
- 16″Strategies
to identify and mitigate risks for first-in-human clinical trials with
investigational medicinal products (CHMP/SWP/28367/07)” (PDF). European Medicines Agency. 1 September 2007. Retrieved 22 January 2016.
- 17 “Accident grave dans le cadre d’un essai clinique – Intervention de Marisol Touraine à Rennes”. Ministère des Affaires Sociales, de la Santé et des Droits des Femmes, France. 15 January 2016.
- 18Barbara Casassus (23 January 2016). “France investigates drug trial disaster” (PDF). The Lancet 387 (10016): 326. doi:10.1016/S0140-6736(16)00154-9.
- 19
- “Expert
reaction to French drug trial – reports of one patient dying and five
others in hospital and of the Paris prosecutor’s office having opened an
investigation into what happened”. Science Media Centre, London. 16 January 2016. Retrieved 21 January 2016.
- 20
- “La
survenue d’effets graves ayant entraîné l’hospitalisation de 6
patients, dont un en état de mort cérébrale, a conduit à l’arrêt
prématuré d’un essai clinique du laboratoire BIAL – Point d’information”. Agence Nationale de Sécurité du Médicament, France (ANSM). 15 January 2016.
- 21
- “Six hospitalized in Bial clinical trial in France”. BioWorld.com. Retrieved 2016-01-17.
- 22
- Enserink M (January 16, 2016). “More details emerge on fateful French drug trial”. Science Magazine. Retrieved January 18, 2016.
- 23
- “France clinical trial: 90 given drug, one man brain-dead”. BBC. January 15, 2016. Retrieved January 16, 2016.
- 24
- “Ce que l’on sait de l’accident survenu lors d’un essai clinique à Rennes”, Le Monde, 15 January 2016
- 25
- Blamont M (January 15, 2016). “French drug trial disaster leaves one brain dead, five injured”. Reuters. Retrieved January 16, 2016.
- 26
- Clarisse Lucas, AFP (January 17, 2016). “Man dies after being left brain-dead in French drug trial”. Yahoo. Retrieved January 17, 2016.
- 27
- “Accident “inédit” lors d’un essai clinique: un homme en état de mort cérébrale, cinq hospitalisés”. La Depeche. January 15, 2016. Retrieved January 18, 2016.
- 28
- “France clinical trial: ‘No known antidote’ to drug”. BBC News. 15 January 2016. Retrieved 18 January 2016.
- 29
- Enserink, Martin (2016). “What We Know So Far About the Clinical Trial Disaster in France” (online). Science (January 15). Retrieved 16 January 2016.
- 30
- “EU Clinical Trials Register”. European Medicines Agency. Retrieved 20 January 2016.
- 31
- “WHO International Clinical Trials Registry Platform”. World Health Organization. Retrieved 20 January 2016.
- 32
- “ANSM – Répertoire public des essais cliniques de médicaments”. Agence Nationale de Sécurité du Médicament et des Produits de Santé, France. Retrieved 20 January 2016.
- 33
- “ClinicalTrials.gov Registry”. U.S. National Institutes of Health. Retrieved 20 January 2016.
- 34
- “Drug trial volunteer dies as ‘manufacturing error’ theory investigated”. The Independent. January 18, 2016. Retrieved January 18, 2016.
- 35
- Chobanian; et al. (10 April 2014). “Discovery of MK-4409, a Novel Oxazole FAAH Inhibitor for the Treatment of Inflammatory and Neuropathic Pain”. ACS Med. Chem. Lett.
- 36
- Merck (15 October 2009). “Merck Pipeline, Oct 2009” (PDF). Merck.
- 37
- “Seven studies found for: jnj-42165279”. Clinicaltrials.gov. Retrieved 2016-01-19.
- 38
- “2 studies found for: V158866”. Clinicaltrials.gov. Retrieved 2016-01-19.
- 39
- Bisogno T, Maccarrone M. Latest advances in the discovery of fatty acid amide hydrolase inhibitors. Expert Opin Drug Discov. 2013 May;8(5):509-22. PMID 23488865 doi: 10.1517/17460441.2013.780021.
- 40
- Shanks KG, Behonick
GS, Dahn T, Terrell A. Identification of novel third-generation
synthetic cannabinoids in products by ultra-performance liquid
chromatography and time-of-flight mass spectrometry. J Anal Toxicol. 2013 Oct;37(8):517-25. PMID 23946450 doi: 10.1093/jat/bkt062.
- 41
- Uchiyama N, Matsuda
S, Kawamura M, Shimokawa Y, Kikura-Hanajiri R, Aritake K, Urade Y, Goda
Y. Characterization of four new designer drugs, 5-chloro-NNEI, NNEI
indazole analog, α-PHPP and α-POP, with 11 newly distributed designer
drugs in illegal products. Forensic Sci Int. 2014 Oct;243:1-13. PMID 24769262 doi: 10.1016/j.forsciint.2014.03.013.
External links
| WO2005073199A1 * |
Jan 15, 2005 |
Aug 11, 2005 |
Aventis Pharma Gmbh |
Indazole derivatives as inhibitors of hormone-sensitive lipases |
| WO2010074588A2 |
Dec 23, 2009 |
Jul 1, 2010 |
BIAL – PORTELA & Cª, S.A. |
Pharmaceutical compounds |
| WO2012015324A1 |
Jul 28, 2011 |
Feb 2, 2012 |
Bial – Portela & Ca, S.A. |
Process for the synthesis of substituted urea compounds |
| US4051252 * |
Nov 24, 1975 |
Sep 27, 1977 |
Bayer Aktiengesellschaft |
3-aminoindazole-1 and 2-carboxylic acid derivatives |
| US4331678 * |
Jan 14, 1980 |
May 25, 1982 |
Fbc Limited |
Carbamoyl pyrazole compounds and their pesticidal application |
| US4973588 * |
Feb 10, 1989 |
Nov 27, 1990 |
Mitsui Petrochemical Industries, Ltd. |
Imidazole derivatives having anti-hypoxia properties |
| US5578627 * |
Oct 27, 1993 |
Nov 26, 1996 |
Toyama Chemical Co., Ltd. |
1,2-benzoisoxazole derivative or its salt and brain-protecting agent comprising the same |
/////////
C1C(CCCC1)N(C)C(=O)n2cc(nc2)c3ccc[n+](c3)O
Share this: