SOVAPREVIR
(2S, 4R) -1 - [(2S)-2-tert-butyl-4-oxo-4-(piperidin-1-yl) butanoyl]-N-{(1R, 2S) -1 - [(cyclopropanesulfonyl) carbamoyl]-2-ethenylcyclopropyl} -4 - [(7-methoxy-2-phenylquinolin-4-yl) oxy] pyrrolidine-2-carboxamide
PATENT
US 2009048297 ENTRY 60
WO 2008008502
CN 103420991
THERAPEUTIC CLAIM ....Treatment of hepatitis C
CHEMICAL NAMES
1. 2-Pyrrolidinecarboxamide, N-[(1R,2S)-1-[[(cyclopropylsulfonyl)amino]carbonyl]-2-
ethenylcyclopropyl]-1-[(2S)-3,3-dimethyl-1-oxo-2-[2-oxo-2-(1-piperidinyl)ethyl]butyl]-4-
[(7-methoxy-2-phenyl-4-quinolinyl)oxy]-, (2S,4R)-
ethenylcyclopropyl]-1-[(2S)-3,3-dimethyl-1-oxo-2-[2-oxo-2-(1-piperidinyl)ethyl]butyl]-4-
[(7-methoxy-2-phenyl-4-quinolinyl)oxy]-, (2S,4R)-
2. (2S,4R)-N-{(1R,2S)-1-[(cyclopropylsulfonyl)carbamoyl]-2-ethenylcyclopropyl}-1-{(2S)-
3,3-dimethyl-2-[2-oxo-2-(piperidin-1-yl)ethyl]butanoyl}-4-[(7-methoxy-2-phenylquinolin-
4-yl)oxy]pyrrolidine-2-carboxamide
3,3-dimethyl-2-[2-oxo-2-(piperidin-1-yl)ethyl]butanoyl}-4-[(7-methoxy-2-phenylquinolin-
4-yl)oxy]pyrrolidine-2-carboxamide
MOLECULAR FORMULA C43H53N5O8S
MOLECULAR WEIGHT 800.0
SPONSOR Achillion Pharmaceuticals, Inc.
CODE DESIGNATION ACH-0141625
CAS REGISTRY NUMBER 1001667-23-7
- ACH-0141625
- Sovaprevir
- UNII-2ND9V3MN6O
Sovaprevir (formerly ACH-0141625), an HCV NS3 protease inhibitor, is in phase II clinical trials at Achillion for the oral treatment of naive patients with chronic hepatitis C virus genotype 1.
In 2012, fast track designation was assigned by the FDA for the treatment of hepatitis C (HCV). In 2013, a clinical hold was placed for the treatment of hepatitis C (HCV) in combination with atazanavir after elevations in liver enzymes associated with the combination of both compounds.
Sovaprevir, previously referred to as ACH-1625, is an investigational, next-generation NS3/4A protease inhibitor discovered by Achillion that is currently on clinical hold. In 2012, Fast Track status was granted by the U.S. Food and Drug Administration (FDA) to sovaprevir for the treatment of chronic hepatitis C viral infection (HCV).
Achillion has initiated a Phase 2 clinical trial (007 Study) to evaluate the all-oral, interferon-free combination of sovaprevir and its second-generation NS5A inhibitor, ACH-3102, with ribavirin (RBV), for a 12 week treatment duration, in treatment naïve, genotype 1 (GT1) HCV patients. In July 2013, sovaprevir was placed on clinical hold after elevated liver enzymes were observed in a Phase 1 healthy subject drug-drug interaction study evaluating the effects of concomitant administration of sovaprevir with ritonavir-boosted atazanavir.
In accordance with the clinical hold, the FDA provided that no new clinical trials that included dosing with sovaprevir could be initiated, however, the FDA allowed continued enrollment and treatment of patients in the Phase 2 -007 clinical trial evaluating 12-weeks of sovaprevir in combination with ACH-3102 and RBV for patients with treatment-naive genotype 1 HCV. In September 2013, after reviewing Achillion's response, the FDA stated that although all issues identified in the June 2013 letter had been addressed, it had concluded that the removal of the clinical hold was not warranted at this time.
The FDA requested, among other things, additional analysis to more fully characterize sovaprevir pharmacokinetics and the intrinsic and extrinsic factors that may lead to higher than anticipated exposures of sovaprevir or other potential toxicities in addition to the observed liver enzyme elevations.
The FDA also requested Achillion's proposed plan for future clinical trials in combination with other directly-acting antivirals. At the request of the FDA, Achillion plans to submit a proposed plan for analyzing the additional clinical, non-clinical and pharmacokinetic data requested before the end of 2013, and if that analysis plan is approved by the FDA, submit a complete response during the first half of 2014. Achillion retains worldwide commercial rights to sovaprevir.
Sovaprevir has demonstrated activity against all HCV genotypes (GT), including equipotent activity against both GT 1a and 1b (IC50 ~ 1nM) in vitro.
With its rapid and extensive partitioning to the liver, as well as high liver/plasma ratios, sovaprevir has been clinically demonstrated to allow for once-daily, non-boosted dosing.
The current safety database for sovaprevir includes more than 560 subjects dosed to date and demonstrates that sovaprevir is well tolerated in these subjects.
Sovaprevir has demonstrated high rates of clinical cures in combination with pegylated-interferon and RBV in a challenging, real world, patient population of genotype 1 treatment-naive patients.
100% of GT1b subjects achieved a rapid virologic response (RVR) in the 007 Study evaluating the interferon-free combination of sovaprevir + ACH-3102 + RBV for 12 weeks. The Phase 2 study is ongoing.
Sovaprevir in vitro retains activity against mutations that confer resistance to 1st-generation protease inhibitors.
In clinical studies to date, sovaprevir has demonstrated a high pharmacologic barrier to resistance with no on-treatment viral breakthrough reported to date in GT1b patients.
Sovaprevir is believed to be synergistic when combined with other classes of DAAs, including the second-generation NS5A inhibitor, ACH-3102.
For more information about the next-generation NS3/4A protease inhibitor, sovaprevir, please see the Related Links on this page or visit Resources.
Sovaprevir is an investigational compound. Its safety and efficacy have not been established. (Updated December 2013)
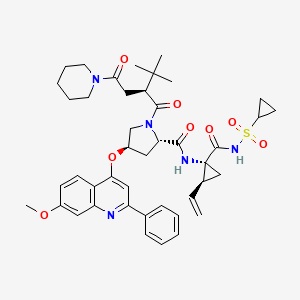
SOVAPREVIR
An estimated 3% of the world's population is infected with the hepatitis C virus. Of those exposed to HCV, 80% become chronically infected, at least 30% develop cirrhosis of the liver and 1-4% develop hepatocellular carcinoma. Hepatitis C Virus (HCV) is one of the most prevalent causes of chronic liver disease in the United States, reportedly accounting for about 15 percent of acute viral hepatitis, 60 to 70 percent of chronic hepatitis, and up to 50 percent of cirrhosis, end-stage liver disease, and liver cancer. Chronic HCV infection is the most common cause of liver transplantation in the U.S., Australia, and most of Europe. Hepatitis C causes an estimated 10,000 to 12,000 deaths annually in the United States. While the acute phase of HCV infection is usually associated with mild symptoms, some evidence suggests that only about 15% to 20% of infected people will clear HCV.
HCV is an enveloped, single-stranded RNA virus that contains a positive-stranded genome of about 9.6 kb. HCV is classified as a member of the Hepacivirus genus of the family Flaviviridae. At least 4 strains of HCV, GT-1-GT-4, have been characterized.
The HCV lifecycle includes entry into host cells; translation of the HCV genome, polyprotein processing, and replicase complex assembly; RNA replication, and virion assembly and release. Translation of the HCV RNA genome yields a more than 3000 amino acid long polyprotein that is processed by at least two cellular and two viral proteases. The HCV polyprotein is:
NH2-C-E1-E2-p7-NS2-NS3-NS4A-NS4B-NS5A-NS5B-COOH.
The cellular signal peptidase and signal peptide peptidase have been reported to be responsible for cleavage of the N-terminal third of the polyprotein (C-E1-E2-p7) from the nonstructural proteins (NS2-NS3-NS4A-NS4B-NS5A-NS5B). The NS2-NS3 protease mediates a first cis cleavage at the NS2-NS3 site. The NS3-NS4A protease then mediates a second cis-cleavage at the NS3-NS4A junction. The NS3-NS4A complex then cleaves at three downstream sites to separate the remaining nonstructural proteins. Accurate processing of the polyprotein is asserted to be essential for forming an active HCV replicase complex.
Once the polyprotein has been cleaved, the replicase complex comprising at least the NS3-NS5B nonstructural proteins assembles. The replicase complex is cytoplasmic and membrane-associated. Major enzymatic activities in the replicase complex include serine protease activity and NTPase helicase activity in NS3, and RNA-dependent RNA polymerase activity of NS5B. In the RNA replication process, a complementary negative strand copy of the genomic RNA is produced. The negative strand copy is used as a template to synthesize additional positive strand genomic RNAs that may participate in translation, replication, packaging, or any combination thereof to produce progeny virus. Assembly of a functional replicase complex has been described as a component of the HCV replication mechanism. Provisional application 60/669,872 “Pharmaceutical Compositions and Methods of Inhibiting HCV Replication” filed Apr. 11, 2005, is hereby incorporated by reference in its entirety for its disclosure related to assembly of the replicase complex.
Current treatment of hepatitis C infection typically includes administration of an interferon, such as pegylated interferon (IFN), in combination with ribavirin. The success of current therapies as measured by sustained virologic response (SVR) depends on the strain of HCV with which the patient is infected and the patient's adherence to the treatment regimen. Only 50% of patients infected with HCV strain GT-1 exhibit a sustained virological response. Direct acting antiviral agents such as ACH-806, VX-950 and NM 283 (prodrug of NM 107) are in clinical development for treatment of chronic HCV. Due to lack of effective therapies for treatment for certain HCV strains and the high mutation rate of HCV, new therapies are needed.
................................................
(2S,4R)-1-((S)-2-tert-butyl-4-oxo-4-(piperidin-1-yl)butanoyl)-N-((1R,2S)-1-(cyclopropylsulfonylcarbamoyl)-2-vinylcyclopropyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-2-carboxamide

SOVAPREVIR IS DESCRIBED AS 60 IN CLAIM
SYNTHESIS OF INTERMEDIATE 13 BELOW AND ALSO COMPD 8 IE SOVAPREVIR IN STEP 4
Example 1
SYNTHESIS OF 1-((2S,4R)-1-((S)-2-TERT-BUTYL-4-OXO-4-(PIPERIDIN-1-YL)BUTANOYL)-4-(7-METHOXY-2-PHENYLQUINOLIN-4-YLOXY)PYRROLIDINE-2-CARBOXAMIDO)-2-VINYLCYCLOPROPANECARBOXYLIC ACID
Step 1. Preparation of N-(cyclopropylsulfonyl)-1-(BOC-amino)-2-vinylcyclopropanecarboxamide
CDI (2.98 g, 18.4 mm, 1.1 eq) is dissolved in ethyl acetate. N-Boc-cyclopropylvinyl acid (3.8 g, 16.7 mm, 1.0 eq), prepared via the procedure given by Beaulieu, P. L. et al. (J. Org. Chem. 70: 5869-79 (2005)) is added to the CDI/ethyl acetate mixture and stirred at RT until the starting material is consumed. Cyclopropyl sulfonamine (2.2 g, 18.4 mm, 1.1 eq) is added to this mixture followed by DBU (2.1 ml, 20.5 mm, 1.23 eq) and the mixture is stirred at RT for 2 h. Workup and purification by silica gel chromatography provides 2g of compound 2.
Step 2. Preparation of (2S,4R)-tert-butyl 2-(1-(cyclopropylsulfonylcarbamoyl)-2-vinylcyclopropylcarbamoyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-1-carboxylate and (2S,4R)—N-(1-(cyclopropylsulfonylcarbamoyl)-2-vinylcyclopropyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-2-carboxamide
Compound 1 (4.3 g, 9.3 mmol, 1.1 eq), prepared according to the method given ins WO 02/060926, in DMF is stirred with O-(Benzotriazol-lyl)-N,N,N′,N′-Tetramethyluronium hexafluorophosphate (4.1 g, 10.5 mmol, 1.3 eq) for 30 minutes, followed by addition of cyclopropylamine 2 (1.92 g, 8.3 mmol, 1.0 eq) and N-methylmorpholine (2.52 g, 25.0 mmol, 3.0 eq). The mixture is stirred over night and the solvent removed under reduced pressure. The resulting residue is diluted with ethyl acetate and washed with saturated aqueous NaHCO3. The organic solvent is dried over MgSO4 and concentrated under reduced pressure to afford crude 3, which is used for next step without further purification.
Compound 3 in 10 ml dry CH2Cl2 is treated with 5 mL TFA and stirred over night. The solvent is removed and the residue recrystallized from ethyl acetate to afford 4.12 g Compound 4 (61% yield two steps).
Step 3. Preparation of (3S)-3-((2S,4R)-2-(1-(cyclopropylsulfonylcarbamoyl)-2-vinylcyclopropylcarbamoyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-1-carbonyl)-4,4-dimethylpentanoic acid
The Acid 5 (58 mg, 0.25 mmol, 1.2 eq), prepared via the procedure given by Evans, D. A., et al. (J. Org. Chem. 64: 6411-6417 (1999)) in 1.2 mL DMF is stirred with 4 (138 mg, 0.21 mmol), HATU (160 mg, 0.42 mmol, 2.0 eq), and DIEA (0.63 mmol, 3.0 eq) overnight. The mixture is subjected to HPLC purification to afford 121 mg 6 (77% yield), which is further treated with 0.5 mL TFA in 1.0 mL DCM overnight. The solvent was removed to provide Compound 7 in 100% yield.
Step 4. Preparation of (2S,4R)-1-((S)-2-tert-butyl-4-oxo-4-(piperidin-1-yl)butanoyl)-N-(1-(cyclopropylsulfonylcarbamoyl)-2-vinylcyclopropyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-2-carboxamide
PLEASE NOTE 8 IS SOVAPREVIR
The Acid 7 (0.15 mmol) in 1.0 mL DMF is stirred with pepridine (excess, 0.6 mmol, 4 eq), HATU (115 mg, 0.3 mmol, 2.0 eq), and DIEA (0.45 mmol, 3.0 eq) for 4 hrs. The mixture is subjected to HPLC purification to afford 77.1 mg 8.
Step 5. Preparation of (3S)-3-((2S,4R)-2-(1-(ethoxycarbonyl)-2-vinylcyclopropylcarbamoyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-1-carbonyl)-4,4-dimethylpentanoic acid
Step 5. Preparation of (3S)-3-((2S,4R)-2-(1-(ethoxycarbonyl)-2-vinylcyclopropylcarbamoyl)-4-(7-methoxy-2-phenylquinolin-4-yloxy)pyrrolidine-1-carbonyl)-4,4-dimethylpentanoic acid
The Acid 5 (105 mg, 0.46 mmol, 1.2 eq) in 1.2 mL DMF is stirred with 9 (202 mg, 0.38 mmol), HATU (290 mg, 0.76 mmol, 2.0 eq), and DIEA (1.2 mmol, 3.0 eq) overnight. The mixture is subjected to HPLC purification to afford 204.3 mg 10 (75% yield), which is further treated with 0.5 mL TFA in 1.0 mL DCM overnight. The solvent is removed to provide 11 in 100% yield.
Step 6. Preparation of Final Product
The Acid 11 (30 mg, 0.045 mmol) in 1.0 mL DMF is stirred with pepridine (0.27 mmol, 6 eq), HATU (34 mg, 0.09 mmol, 2.0 eq), and DIEA (0.14 mmol, 3.0 eq) for 2 hrs. The mixture is subjected to HPLC purification to afford 21.2 mg 12 (65% yield), which is hydrolyzed in methanol with 2N NaOH for 6 hrs. The mixture is acidified with 6N HCl and subjected to HPLC purification to afford 7.6 mg 13.
....................................


THANKS AND REGARD'S
DR ANTHONY MELVIN CRASTO Ph.D
DR ANTHONY MELVIN CRASTO Ph.D
GLENMARK SCIENTIST , NAVIMUMBAI, INDIA
did you feel happy, a head to toe paralysed man's soul in action for you round the clock
need help, email or call me
MOBILE-+91 9323115463
web link
I was paralysed in dec2007, Posts dedicated to my family, my organisation Glenmark, Your readership keeps me going and brings smiles to my family






No comments:
Post a Comment